- Posts: 54
 MARTINI simulation of protein-protein assosiation
MARTINI simulation of protein-protein assosiation
- James Starlight
-
 Topic Author
Topic Author
- Visitor
I am looking for some tutorial which will be usefull for the MARTINI simulation of
protein-protein association dealing with system consisted of two separate macrompolecules in unbound form (E.g placed on some distance between each other):
1) big membrane protein embedded within the membrane
2) the small water-soluble protein.
Assuming that I have successfully simulated in MARTINI MDs: 1 and 2 separately and now would like to reproduce its association starting from those 2 proteins in the unbound form.
Because existing tutorials consisted of only the information regarding assembly of single protein (or bound complex) the question: how I can prepare such complex system consisted of big protein in membrane plus water soluble protein (e.g placed in water within the 1 nm distance from the bigger protein)?
Thanks in advance for help!
Please Log in or Create an account to join the conversation.
- flaviyan
-

- Offline
- Senior Boarder
The DAFT approach will be your best help on this. It is designed to study protein protein interaction. It is highlights in the obligations section. And the tutorials is hosted at www.biotechnik.nat.uni-erlangen.de/resea...ads/DAFT/index.shtml
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
regarding construction of the system consisted of the both membrane protein within the membrane and protein within the water
is it simpler possible to use algorithm how we put the protein within the pre equilibrated membrane
1) use CG structure of the protein within the membrane obtained from the first run of the insane and then 2) put second protein within it water part again using insance.py *here I don't know how to provide both gro files as the inputs) with some additional options e.g to orient the second protein along Z and shift it from the membrane on that principal axe on the chosen distance placing thus in water. Or alternatively use here genbox in the following manner
g_genbox -cp water-soluble-prot.pdb -cs membrane-system.gro -vdwd 0.21 -o solvated.gro
Will be thankful for any suggestions regarding realization of such work flow!
J.
Please Log in or Create an account to join the conversation.
- Pim
-

- Offline
- Expert Boarder
- Posts: 105
If you have GROMACS 5 available, you can use gmx insert-molecules -ci water-soluble-prot.pdb -cp membrane-system.gro -nmol 1 -ip positions.dat -rot xyz
where positions.dat contains the x,y and z distance the molecule needs to be shifted from the coordinates in the pdb file and -rot xyz makes sure it is rotated randomly.
Otherwise, I think it is also possible with insane.py if you run it twice, the second time using flag -rotate random -dm "z" to move the soluble protein along the z-axis.
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
g_genbox -cp waterSOLprot.gro -cs MEMBRANEsystem.gro -vdwd 0.21 -o solvatedNEW.gro -box 18 18 28
than checking VMD everything is looks perfect- the waterSOLprot has been placed in proper position and overlapped CG water has been removed.
now running gromppt I received
WARNING 1 : 2886 non-matching atom names atom names from system.top will be used atom names from system.gro will be ignored I also should specify that after genbox I edited new topology.top manually putting chains of the proteins in correct order (like in new gro file produced by genbox) and putting correct number of W assuming that initially I have multi chain protein in the POPC membrane and using genbox I added to the system one new chain Z before [quote]Protein_A 1 Protein_B 1 Protein_C 1 Protein_D 1 Protein_E 1 Protein_F 1 Protein_G 1 Protein_H 1 Protein_I 1 Protein_J 1 Protein_K 1 Protein_L 1 Protein_M 1 POPC 451 CHOL 0 POPC 451 CHOL 0 W 61226 NA+ 678 CL- 671[/quote] after [quote]Protein_Z 1 Protein_A 1 Protein_B 1 Protein_C 1 Protein_D 1 Protein_E 1 Protein_F 1 Protein_G 1 Protein_H 1 Protein_I 1 Protein_J 1 Protein_K 1 Protein_L 1 Protein_M 1 POPC 451 CHOL 0 POPC 451 CHOL 0 W 60817 NA+ 678 CL- 671[/quote] where I did mistake?[file system.top, line 49]:
2886 non-matching atom names
atom names from system.top will be used
atom names from system.gro will be ignored
I also should specify that after genbox I edited new topology.top manually
putting chains of the proteins in correct order (like in new gro file produced by genbox) and putting correct number of W
assuming that initially I have multi chain protein in the POPC membrane
and using genbox I added to the system one new chain Z
before
Protein_A 1
Protein_B 1
Protein_C 1
Protein_D 1
Protein_E 1
Protein_F 1
Protein_G 1
Protein_H 1
Protein_I 1
Protein_J 1
Protein_K 1
Protein_L 1
Protein_M 1
POPC 451
CHOL 0
POPC 451
CHOL 0
W 61226
NA+ 678
CL- 671
after
Protein_Z 1
Protein_A 1
Protein_B 1
Protein_C 1
Protein_D 1
Protein_E 1
Protein_F 1
Protein_G 1
Protein_H 1
Protein_I 1
Protein_J 1
Protein_K 1
Protein_L 1
Protein_M 1
POPC 451
CHOL 0
POPC 451
CHOL 0
W 60817
NA+ 678
CL- 671
where I did mistake?
Please Log in or Create an account to join the conversation.
- Pim
-

- Offline
- Expert Boarder
- Posts: 105
James Starlight wrote: btw I have tried to insert second molecular using just genbox having both components oriented properly in the space
g_genbox -cp waterSOLprot.gro -cs MEMBRANEsystem.gro -vdwd 0.21 -o solvatedNEW.gro -box 18 18 28
than checking VMD everything is looks perfect- the waterSOLprot has been placed in proper position and overlapped CG water has been remomed.
It's a bit unusual to do it that way. I'd first make the membrane system and the protein separately without solvent, then use
genbox -cp MEMBRANEsystem.gro -ci waterSOLprot.gro -o out.gro -nmol 1 -vdwd 1.2 (or whatever you'd like your minimum distance between you soluble protein and your membrane want to be)
and then us genbox again to solvate
genbox -cp out.gro -cs water.gro -vdwd 0.21 -o solvated.gro
Nonetheless if your way works indeed your membrane system will be below the soluble protein in the gro file. You indeed need to get the top file in the right order. grompp will tell you which atom names are wrong, you'll need to debug from there I'm afraid, I don't see anything wrong with what you've shown here.
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
the protein which was embedded in the membrane here used as the solvent in my case >)
I fix an issue by means of the reorientation of the atom names by hands (copy protein and lipids from the initial pdbs which were submitted to genbox to the
complex.gro obained aftyer its processing) - because initially I had properly oriented complexes in 3d space and I used genbox only to remove overlapped water between second protein and water from the first complex.
Now mdrun works OK!
Of course it will be cool if insane can do all of that staff automatically in 1 step!
BTW
where I can find infroregarding mdp options for the system like mine
which include 2 unbound protein complexes.
E.g the question- how it better to define coupling groups for the barostat and thermostat assuming that 1 protein is inserted within the membrane and another is within the water.
Thanks!
J
Please Log in or Create an account to join the conversation.
- Pim
-

- Offline
- Expert Boarder
- Posts: 105
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
Does merging of the protein and lipids within common group will be not good in my case in opposite to md of the protein embedded within the lipids?
What T coupling constat should be better for all of three groups assuming that I am using berebdsen thermostat in both production and equilibration runs? Does the switching to V-rescale might improve smth?
Thanks again!
J.
Please Log in or Create an account to join the conversation.
- Pim
-

- Offline
- Expert Boarder
- Posts: 105
You can find some tips on the latest mdp parameters here: cgmartini.nl/index.php/mdp
The velocity rescale thermostat is in principle more realistic and doesn't cost you much. I'd recommend it if you're still starting out your project. As the mdp files in the link will tell you, typically 1 ps is used for the constants. Note that the P-R barostat will only work properly if you're system has been pre-equilibrated (as gromacs will suggest if you try).
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
Btw what this warning tell us about and how it could be avoided? Here It referred to the water soluble protein which is chain-z in my system:
WARNING 1 : The bond in molecule-type Protein_Z between atoms 1 BB and 2 BB has an estimated oscillational period of 9.7e-02 ps, which is less than 5 times the time step of 2.0e-02 ps. Maybe you forgot to change the constraints mdp option.[file system.top, line 50]:
The bond in molecule-type Protein_Z between atoms 1 BB and 2 BB has an
estimated oscillational period of 9.7e-02 ps, which is less than 5 times
the time step of 2.0e-02 ps.
Maybe you forgot to change the constraints mdp option.
Please Log in or Create an account to join the conversation.
- flaviyan
-

- Offline
- Senior Boarder
- Posts: 54
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
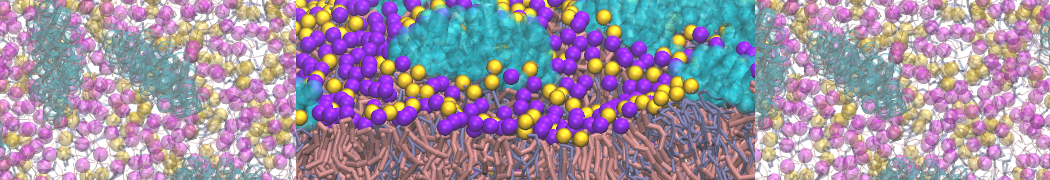
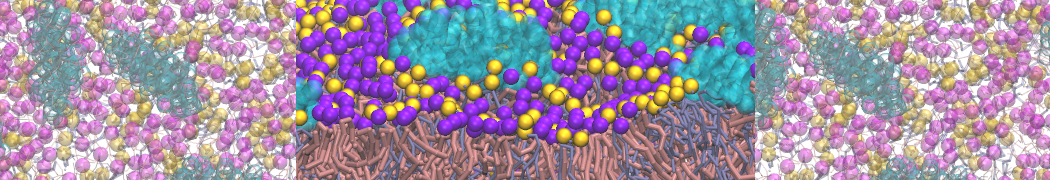
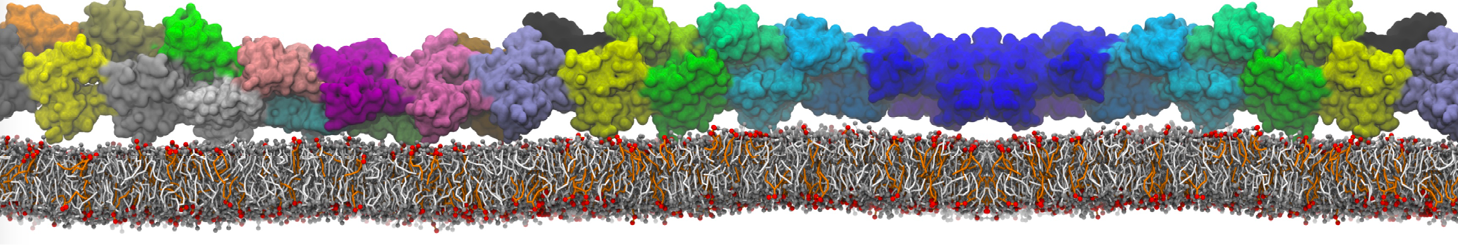
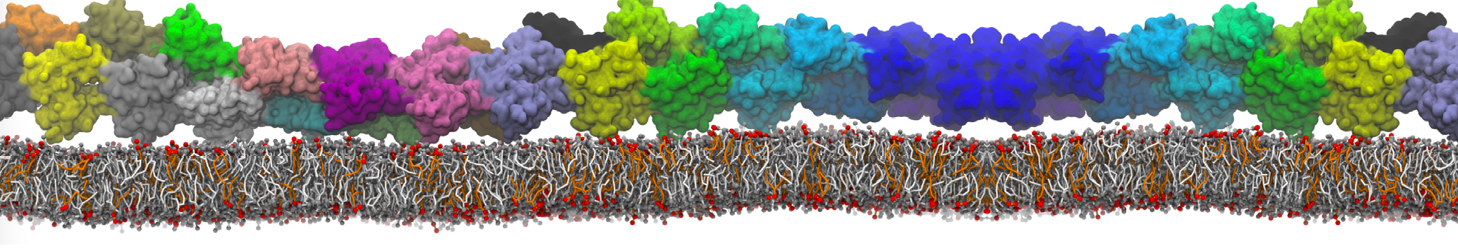
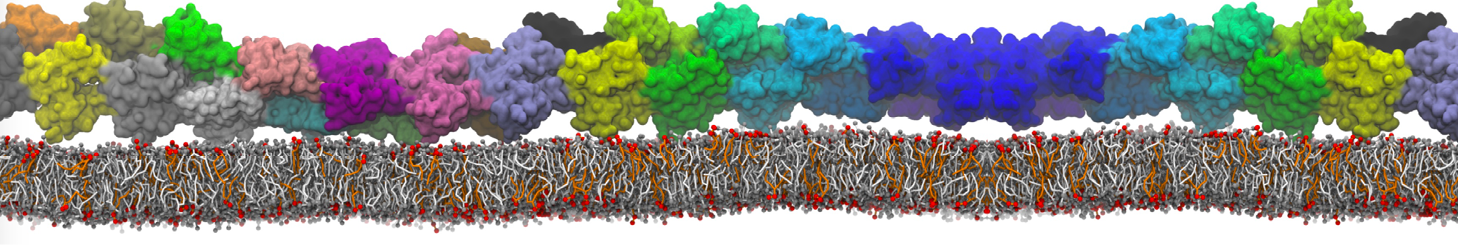
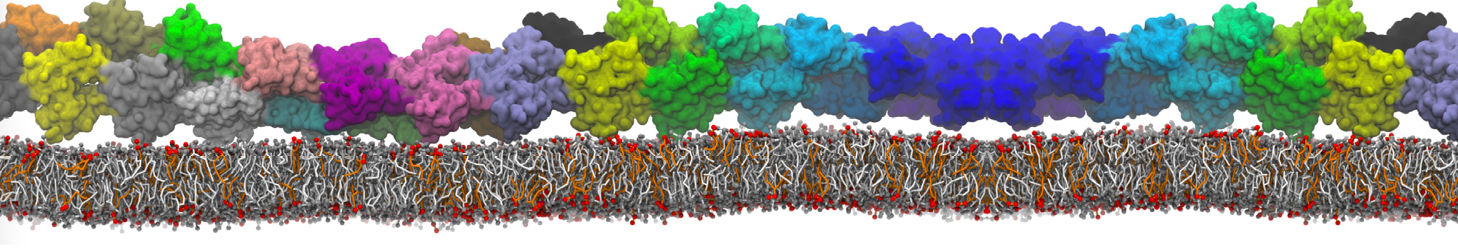
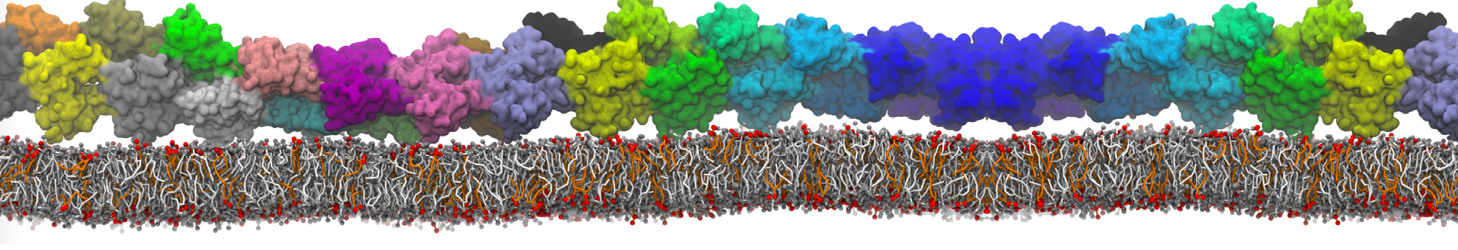
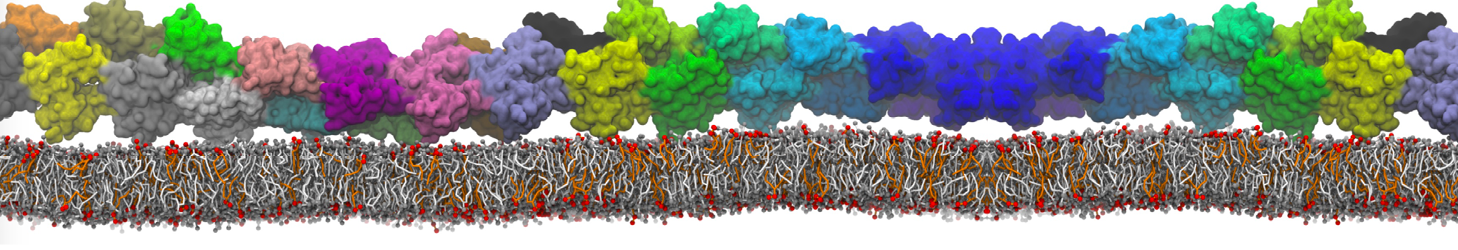
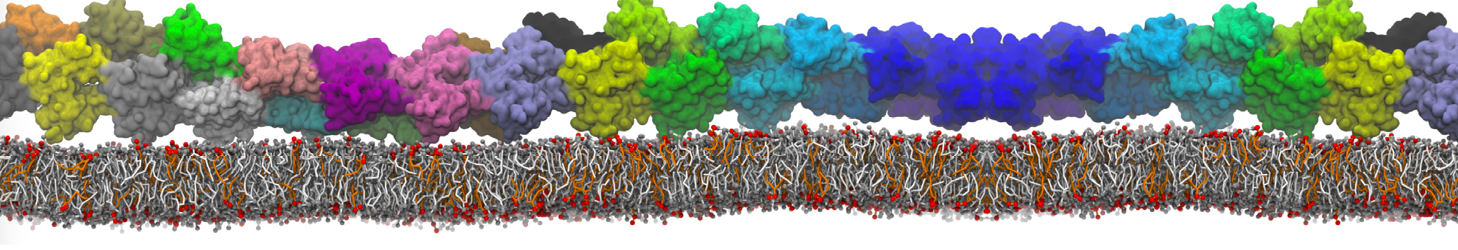
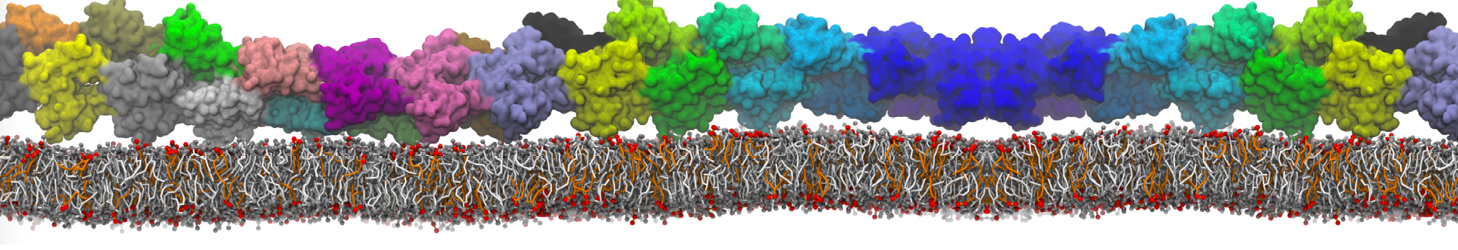
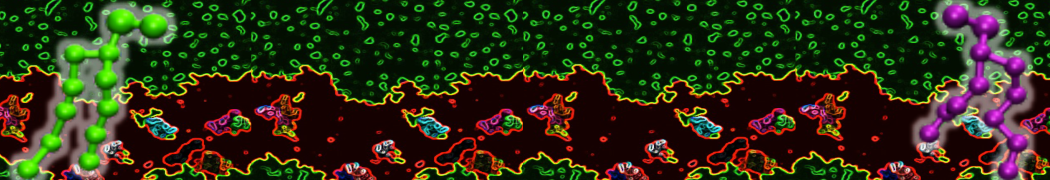
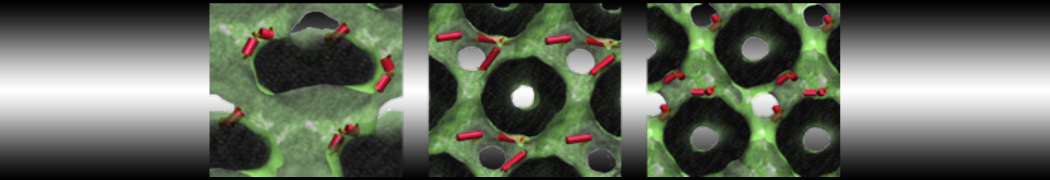
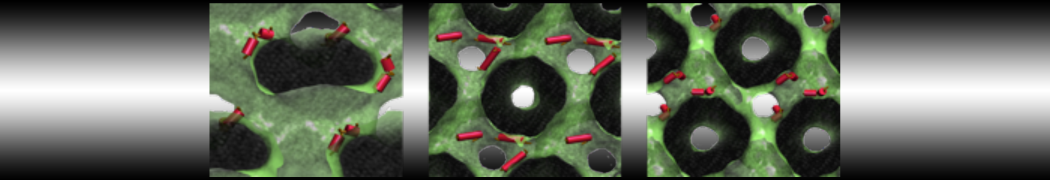
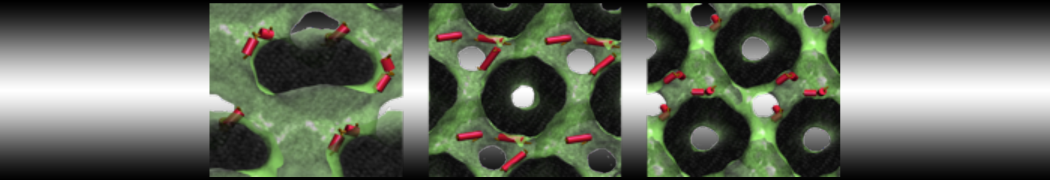
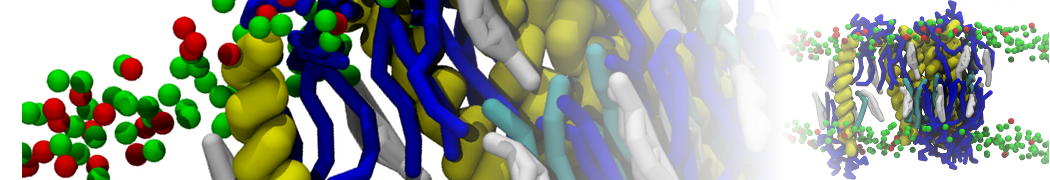
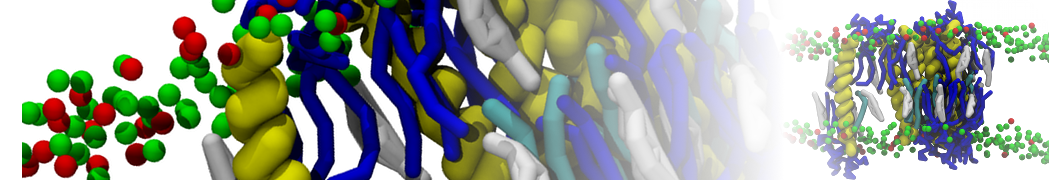
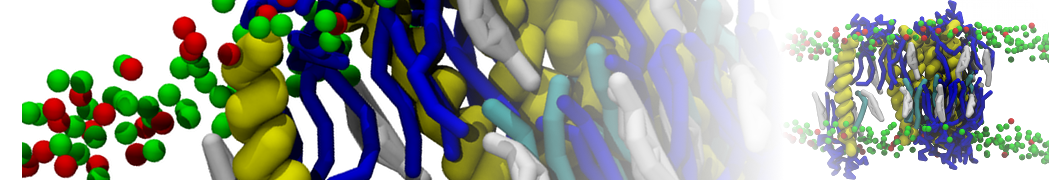
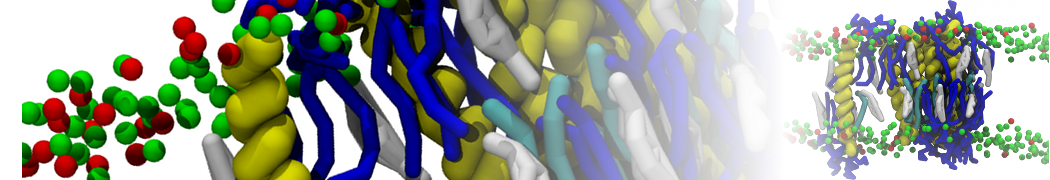
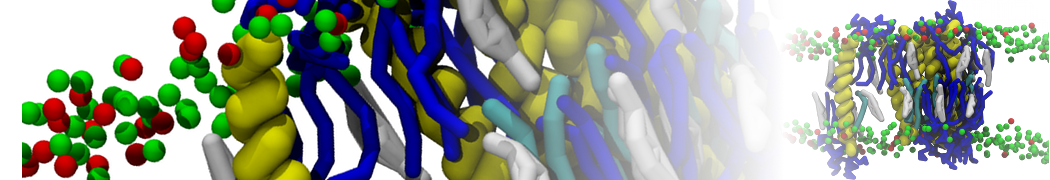
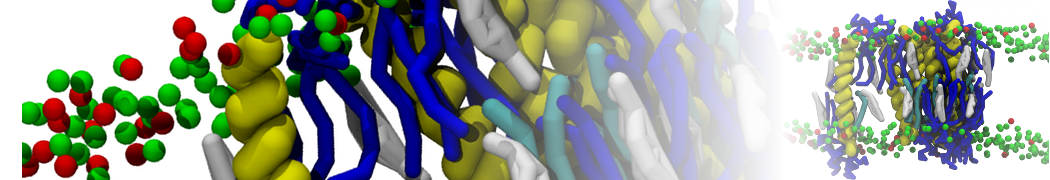
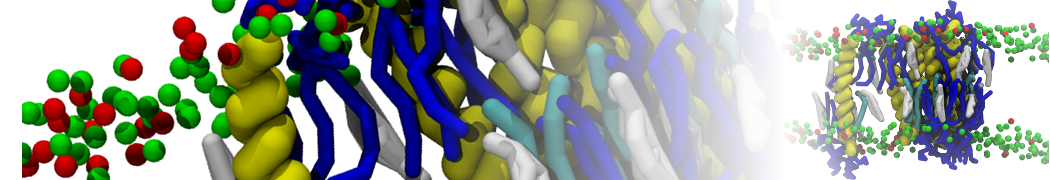
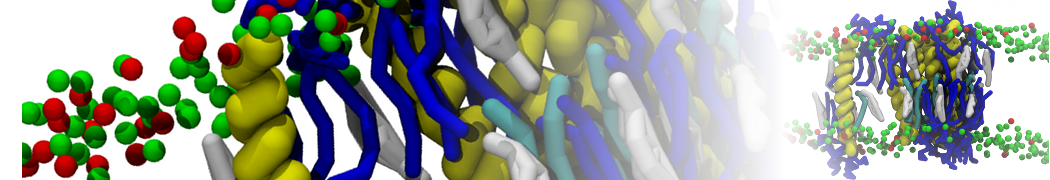
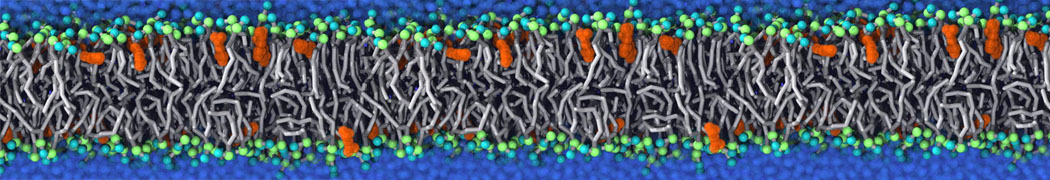
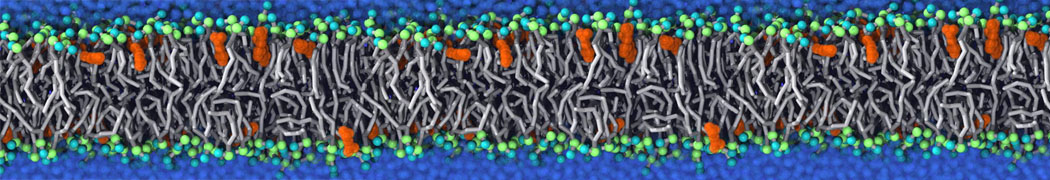
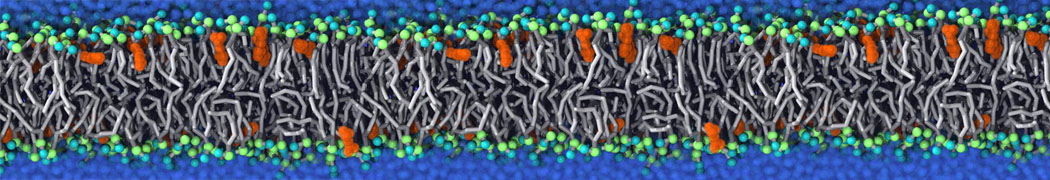
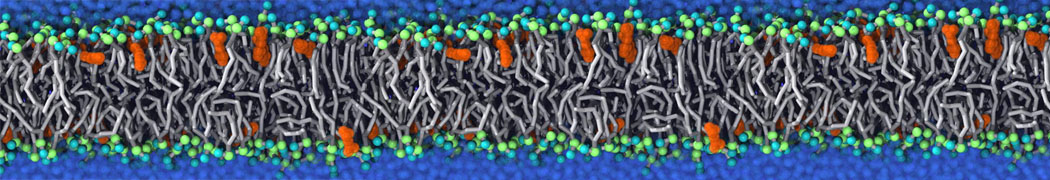
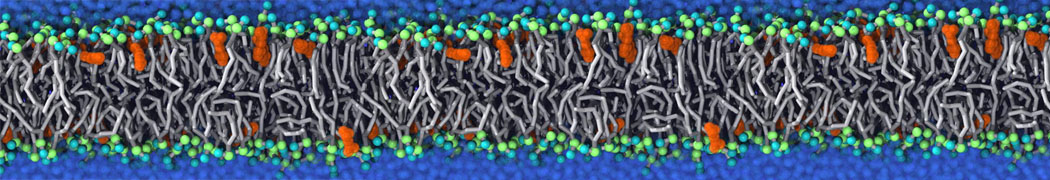
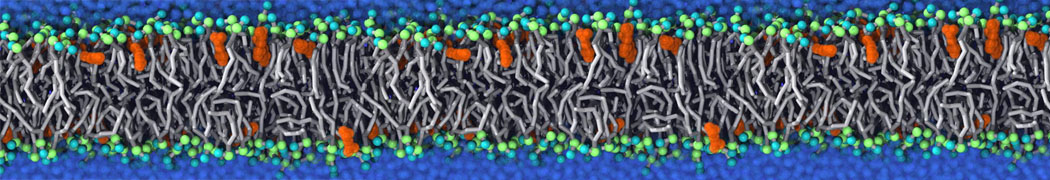
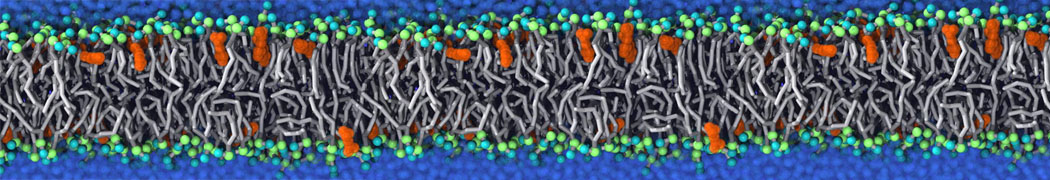
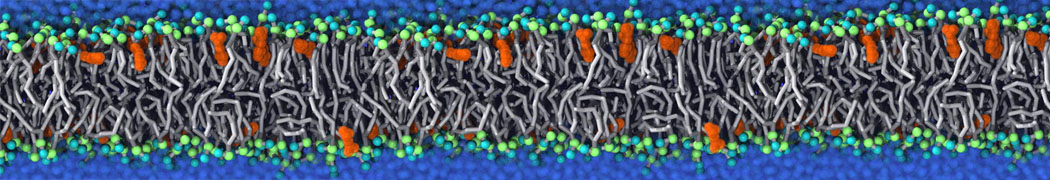
during md run I am interested to simulate binding between part of the membrane protein exposed to water as well as whole smaller water soluble protein.
Understanding of my system could be taken from that short video which is 100ns unrestrained equilibration, from that trajectory I am going to take several snapshots corresponded to different position of wsp (red) relatively memrabrane exposed part and performs 5-10 independent unrestrained MDs to see where it will converges in terms of binding interface:
youtu.be/9_XVvXUMDPY
Assuming that the first recognition between both guys should be mediated (at least at some distance between them) by the electrostatic forces
1 - on what minimum distance should I put both proteins in the system to exclude some bias just from the begining of the MDs assuming that water exposed surface of membrane guy can generate strong local electric field (there are also around 450 ions in this system to compensate charge according to the physiological salt concentration used by default in this model)
2- how I can modify cut-offs (for the Gromacs 4.5) to play with it more
Below is how I using it right now taken from the default Martini mdp
coulombtype = Shift
rcoulomb_switch = 0.0
rcoulomb = 1.2
epsilon_r = 15
vdw_type = Shift
rvdw_switch = 0.9
rvdw = 1.2
DispCorr = No
J.
Please Log in or Create an account to join the conversation.
- flaviyan
-

- Offline
- Senior Boarder
- Posts: 54
Answer to question 1:
I would recommend that the minimum bead-bead distance in any direction to be twice the distance of the cut-off + 1 nm. i.e 2.4 nm + 1 nm.
I am not sure about the second question, but I would not recommend changing these cut-off, unless you are using the Verlet cut-off scheme, in which case you can set the cut-off to 1.1 nm. refer to the new-rf.mdp
Please Log in or Create an account to join the conversation.
- peterkroon
-

- Offline
- Gold Boarder
- Posts: 210
changing the cutoffs will invalidate the forcefield. So don't do that ;)
As an side note: if you are interested in electrostatic interactions, be sure to use the polarizable water model; you may also want to look at the 2.2P model, but refer to the paper to see what it's exact benefits are.
Peter
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
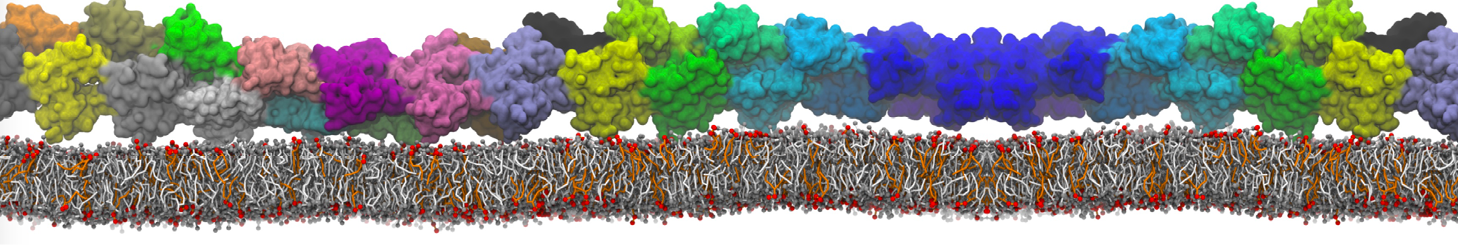
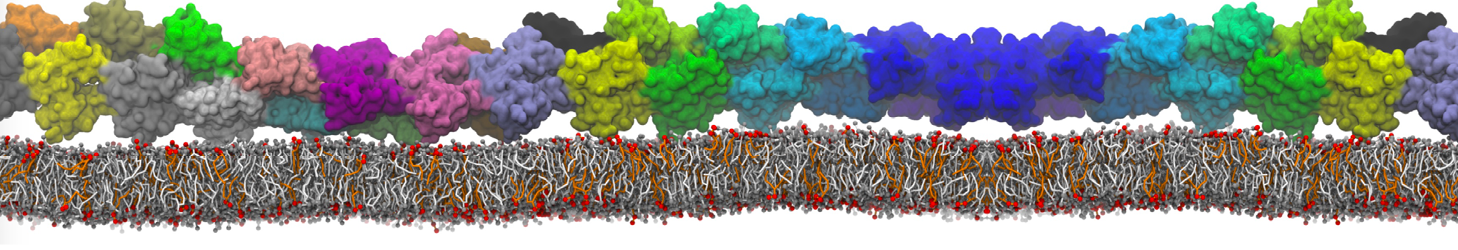
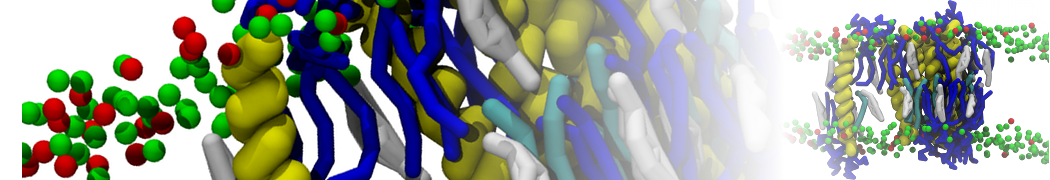
I am looking for some non-complicated analysis of protein-protein association during several MD simulations.
Firstly using my MARTINI model I have simulated association between water soluble protein and some water-exposed part of the membrane protein mediated mainly by the electrostatics attraction between both. Now assuming that I have collected 10 trajectories corresponded to 10 independent MD runs of that process I am looking for some analysis method e.g to cluster the binding interfaces occurred during those runs. to characterize precisely residues which are crucial for binding etc. Will be thankful for any suggestions regarding realization assuming that in my model I have big membrane protein consisted of 12 independent chain in Gromacs topology and small water soluble protein defined as independent chain. The brief vizualisation of that I am looking to analyze is available here youtu.be/u3vKKBq4G6s :-)
Thanks for the help!
J.
Please Log in or Create an account to join the conversation.
- flaviyan
-

- Offline
- Senior Boarder
- Posts: 54
The answer to this question depends on what are your expectation when you performed these simulations?
if you are looking for a binding sites and would like to quantify their relative energy with one another then 10 run might not be enough to see any convergence. Again refer to the DAFT analysis paper pubs.acs.org/doi/pdf/10.1021/ct5010092 that has given a quantitative frame work for such analysis.
If you are only interested in different binding sites then combine your trajectories together and center your receptor protein all these trajectories (make sure the orientation is correct in each of them, refer to the trjconv options in gromacs) then calculate the density map using the gmx densmap tool of your ligand. You will see the possible binding sites.
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
BTW any other methods worked on merged trajectories will work here as well e,g covariance-maxtrix based?
Thanks again!
J.
Please Log in or Create an account to join the conversation.
- flaviyan
-

- Offline
- Senior Boarder
- Posts: 54
Please Log in or Create an account to join the conversation.