- Posts: 2
 troubles with protein-dppc interaction
troubles with protein-dppc interaction
- blanco
-
 Topic Author
Topic Author
- Offline
- Fresh Boarder
Less
More
10 years 7 months ago - 10 years 6 months ago #2086
by blanco
troubles with protein-dppc interaction was created by blanco
Hi,
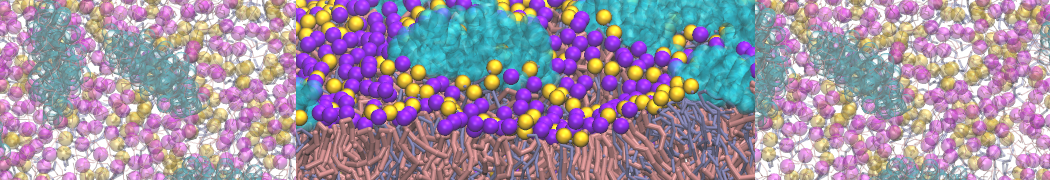
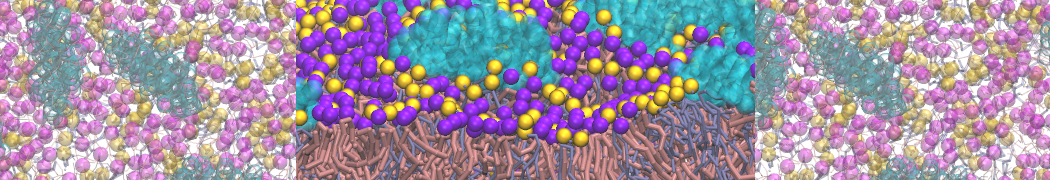
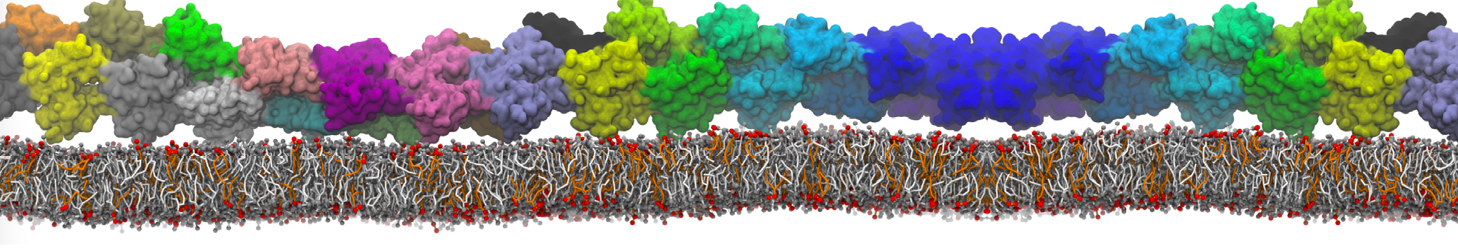
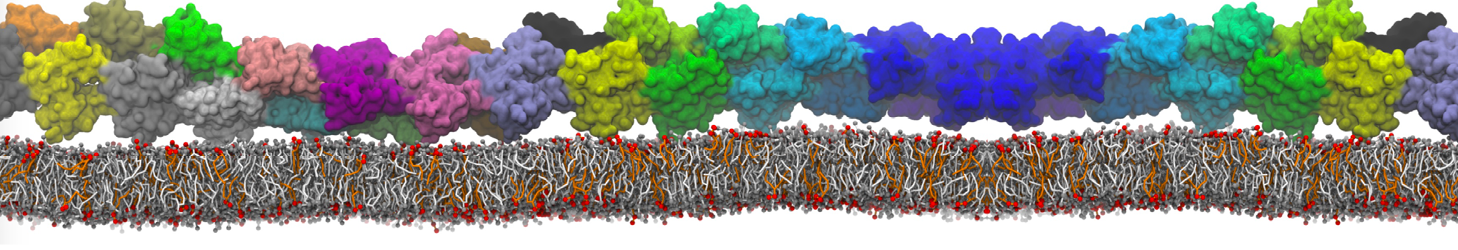
I'm having troubles to reproduce interaction between a charged peptide and a DPPC bilayer. I created the peptide's topology from an atomistic structure with martinize.py script selecting -ff martini22p. I'm using polarizable water (with and without PME). In atomistic resolution the peptide bind strongly to the membrane's surface but this is not the case with the CG model, the dppc-protein LJ and coulombic energy terms fluctuate to negative values when the peptide is close the membrane's head-groups but never bind to them as in the AA model.
The coulombic interactions seems to be not strong enough.
Is this a limitation proper of a CG model, or I am doing something wrong?
What about mixing CG (for bilayer and water) and atomistic (for peptide) models?
Any help would be very appreciated
thanks in advance
regards,
I'm having troubles to reproduce interaction between a charged peptide and a DPPC bilayer. I created the peptide's topology from an atomistic structure with martinize.py script selecting -ff martini22p. I'm using polarizable water (with and without PME). In atomistic resolution the peptide bind strongly to the membrane's surface but this is not the case with the CG model, the dppc-protein LJ and coulombic energy terms fluctuate to negative values when the peptide is close the membrane's head-groups but never bind to them as in the AA model.
The coulombic interactions seems to be not strong enough.
Is this a limitation proper of a CG model, or I am doing something wrong?
What about mixing CG (for bilayer and water) and atomistic (for peptide) models?
Any help would be very appreciated
thanks in advance
regards,
Last edit: 10 years 6 months ago by blanco. Reason: expand discussion
Please Log in or Create an account to join the conversation.
- djurre
-

- Offline
- Admin
Less
More
- Posts: 272
10 years 6 months ago #2297
by djurre
Replied by djurre on topic troubles with protein-dppc interaction
It could be a limitation of the CG-model. However, I think people in general see adhesion of different molecules to membranes. Also since the DPPC is net neutral, I'm suprised the coulombic interactions are so important.
Regarding mixing CG and atomistic: that would be super cool, but I don't think anybody knows exactly how to do it. See also:
T.A. Wassenaar, H. Ingolfsson, M. Prieß, S.J. Marrink, L.V. Schäfer. Mixing Martini: electrostatic coupling in hybrid atomistic – coarse-grained biomolecular simulations. J. Phys. Chem. B, , 117:3516–3530, 2013. pubs.acs.org/doi/abs/10.1021/jp311533p
Regarding mixing CG and atomistic: that would be super cool, but I don't think anybody knows exactly how to do it. See also:
T.A. Wassenaar, H. Ingolfsson, M. Prieß, S.J. Marrink, L.V. Schäfer. Mixing Martini: electrostatic coupling in hybrid atomistic – coarse-grained biomolecular simulations. J. Phys. Chem. B, , 117:3516–3530, 2013. pubs.acs.org/doi/abs/10.1021/jp311533p
Please Log in or Create an account to join the conversation.
- siewert
-

- Offline
- Admin
Less
More
- Posts: 61
10 years 6 months ago #2298
by siewert
Replied by siewert on topic troubles with protein-dppc interaction
Indeed it might be a shortcoming of Martini - we have observed similar issues with charged peptides.
I assume your peptides carry positive charges (Lys, Arg)? In that case a small energetic barrier exists for entering of these side chains into the bilayer, see Fig. 1 in [D.H. de Jong, G. Singh, W.F.D. Bennett, C. Arnarez, T.A. Wassenaar, L.V. Schäfer, X. Periole, D.P. Tieleman, S.J. Marrink. Improved parameters for the Martini coarse-grained protein force field, JCTC, 9:687–697, 2013]. Summed over several of these residues, this barrier might prevent peptides from easily going into the membrane. I am sure they would eventually enter (or remain at the interface when you put them there).
To improve the model, you might play with the Qd interactions, either reducing the Qd-P4 interaction (making Lys and Arg less soluble in water), or increase the strength of the Qd-No interaction (attracting the peptides toward the lipid glycerol region).
Please provide us with feedback about the outcome of these tests, in case you are going to explore along these lines!
I assume your peptides carry positive charges (Lys, Arg)? In that case a small energetic barrier exists for entering of these side chains into the bilayer, see Fig. 1 in [D.H. de Jong, G. Singh, W.F.D. Bennett, C. Arnarez, T.A. Wassenaar, L.V. Schäfer, X. Periole, D.P. Tieleman, S.J. Marrink. Improved parameters for the Martini coarse-grained protein force field, JCTC, 9:687–697, 2013]. Summed over several of these residues, this barrier might prevent peptides from easily going into the membrane. I am sure they would eventually enter (or remain at the interface when you put them there).
To improve the model, you might play with the Qd interactions, either reducing the Qd-P4 interaction (making Lys and Arg less soluble in water), or increase the strength of the Qd-No interaction (attracting the peptides toward the lipid glycerol region).
Please provide us with feedback about the outcome of these tests, in case you are going to explore along these lines!
Please Log in or Create an account to join the conversation.
- blanco
-
 Topic Author
Topic Author
- Offline
- Fresh Boarder
Less
More
- Posts: 2
10 years 6 months ago #2349
by blanco
Replied by blanco on topic troubles with protein-dppc interaction
Thanks both of you. I will do the tests
Please Log in or Create an account to join the conversation.
Time to create page: 0.105 seconds