- Posts: 54
 MARTINI simulation of protein-protein assosiation
MARTINI simulation of protein-protein assosiation
- James Starlight
-
 Topic Author
Topic Author
- Visitor
1- Might the APBS method implemented in VMD be useful for the realization of this task ?
2- What another analysis routines might be apply onto the CG trajectory for the analysis of protein-protein association especially including long-ranged effects?
J.
Please Log in or Create an account to join the conversation.
- flaviyan
-

- Offline
- Senior Boarder
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
as I noticed in my Martini model the order of residues is altered compared to original pdb from which this model has been martimized. E.g in case of multi-chain protein my full atomistic pdb has 10 chain arranged in order like
A: 1-100
B: 1-200
C: 1-47
etc
but in the final MARTINI model its order is altered: all residues now are numbered in the straight manner- from 1 to 347 in the upper case and the chain order now is shifted on one letter in advance.
Are there some trivial way to restore in martini trajectory the order of residues like from the initial pdb e.g using back-mapping (providing topology of the full atomistic pdb as the input to CG topology and trajectory with proper atom order)?
Thanks!
J.
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
assuming I extracted from the CG trajectory several snapshots (as separate pdbs) of the most interests - where the complex between A and B was established.
Now I would like to convert those snapshots to FG representation for some analysis e.g APBS.
How to do it in most trivial way assuming that I have tpr for CG model and can make it for the full atomic pdb of the same system using pdb2gmx for instance.
J
Please Log in or Create an account to join the conversation.
- flaviyan
-

- Offline
- Senior Boarder
- Posts: 54
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
However briefly for my task (CG to FG conversion of the several pdb files) what will be easy to do back-mapping or reverse-transformation ?
Thanks!
Gleb
Please Log in or Create an account to join the conversation.
- flaviyan
-

- Offline
- Senior Boarder
- Posts: 54
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
Back-mapping has worked perfectly in my case :-)
J.
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
assuming long-ranged electrostatics is important for the recognition of two proteins called A and B in my martini system. Assuming that I put both proteins on the distance bigger than electrostatics cut-offs (in my system it's 5-7 nm) does it means that in my case long-range effects are not considered at all?
If so, can I alternatively increase cutoffs, epsilon_r or smth another via modification of the default params used for GMX 4.5
rlist = 1.2
coulombtype = Shift
rcoulomb_switch = 0.0
rcoulomb = 1.2
epsilon_r = 15
vdw_type = Shift
rvdw_switch = 0.9
rvdw = 1.2
DispCorr = No
if it's not possible- does the simulation on GPU (where cut-offs are infinitive) produce better results for my case?
Thanks!
J,
Please Log in or Create an account to join the conversation.
- riccardo
-

- Offline
- Platinum Boarder
Yes, you don't have long-range electrostatics. However, you cannot arbitrarily increase cut-offs. If you believe long-range electrostatics may play a role, you could test this by using the polarizable version of Martini, using PME (see journals.plos.org/ploscompbiol/article?i...journal.pcbi.1000810 ).
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
However in my case (not polarize ff) in 80% of cases (8 runs from 10) the smaller water soluble protein find the bigger one (membrane receptor) very quick being located initially on 10 A distance await from it and establish with it strong contact kept complex until the MD!
Could we assume in this case that just rot-trans diffusion of water-soluble protein is the only what is responsible for the recognition? and the robust of complex is mainly due to the short-ranged effect (vdw strong attraction and local salt-bridges established upon binding).
Is it reasonable to track that hypothesis further to perform simulations with polarize water and compare the results?
Thanks for help!
J.
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
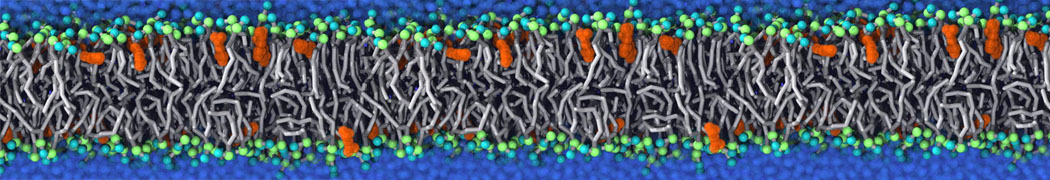
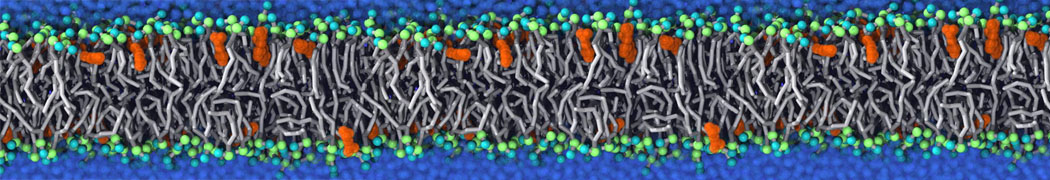
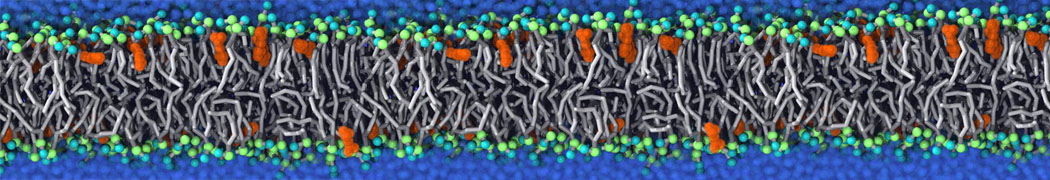
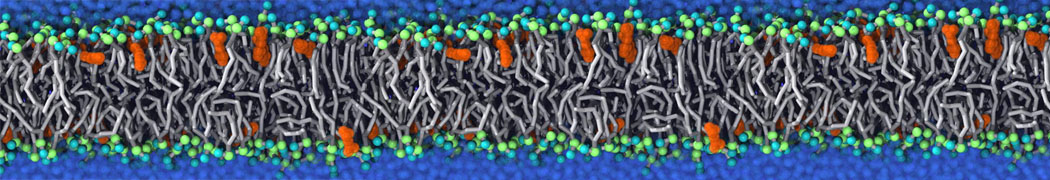
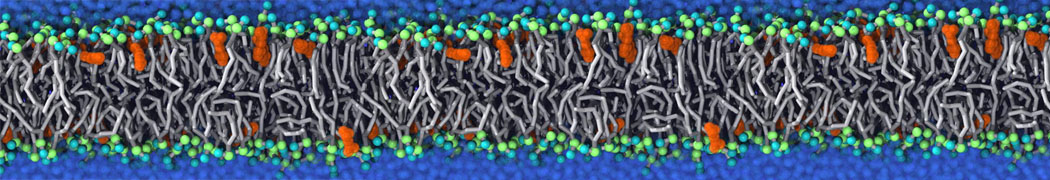
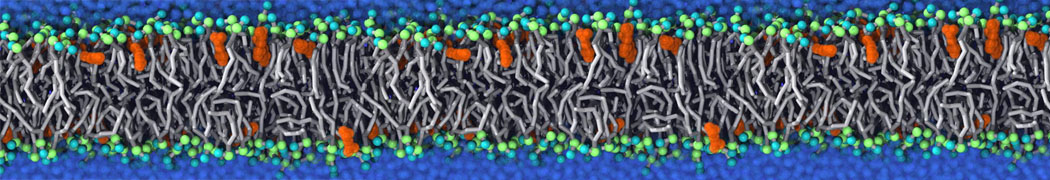
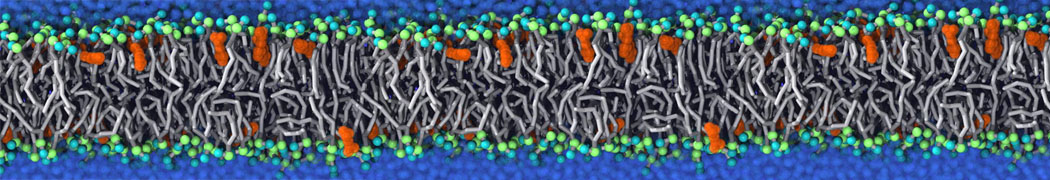
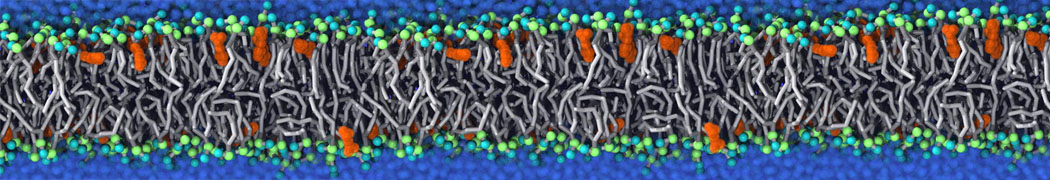
Will it sufficient in case of simulation of membrane protein to use elnedyn2.2p force field during the parametrization of the protein
python ./input/martinize.py -f ./tmp/combined_Z.pdb -o topol_cg.top -dssp /projects/clouddyn/Software/dssp -p backbone -ff elnedyn22p -x CG.pdb
as well as polarize water model PW during solvation
./input/insane.py -f CG.pdb -o system2.gro -p system2.top -pbc cubic -box 18,18,25 -l POPC:4 -l CHOL:0 -salt 0.15 -center -sol PW
Should also something be modified in mdp ?
E.g grompp sent me the following warnings during equilibration of the minimized system
WARNING 1 : The bond in molecule-type Protein_A between atoms 1 BB and 2 BB has an estimated oscillational period of 9.7e-02 ps, which is less than 5 times the time step of 2.0e-02 ps. Maybe you forgot to change the constraints mdp option. WARNING 2 [file ./mdp/equil.mdp]: For proper integration of the Berendsen thermostat, tau_t (0.5) should be at least 5 times larger than nsttcouple*dt (0.2)[/quote] Thanks! Gleb[file system.top, line 26]:
The bond in molecule-type Protein_A between atoms 1 BB and 2 BB has an
estimated oscillational period of 9.7e-02 ps, which is less than 5 times
the time step of 2.0e-02 ps.
Maybe you forgot to change the constraints mdp option.
WARNING 2 : For proper integration of the Berendsen thermostat, tau_t (0.5) should be at least 5 times larger than nsttcouple*dt (0.2)[/quote] Thanks! Gleb[file ./mdp/equil.mdp]:
For proper integration of the Berendsen thermostat, tau_t (0.5) should be
at least 5 times larger than nsttcouple*dt (0.2)
Thanks!
Gleb
Please Log in or Create an account to join the conversation.
- paulocts
-

- Offline
- Fresh Boarder
- Posts: 9
About the mdp: take a look in this examples:
md.chem.rug.nl/index.php/force-field-par...ers/input-parameters
The commentaries of them explain very well what you should change in you mdp when you use polarizable water model. Basically, you should change your relative electrostatic screening (epsilon_r = 2.5) and maybe the Coulomb-type potential (coulombtype = PME). The use of PME should be more realistic than Shift potential in your case .
If you have a lot of lincs warnings, you can use lincs_warnangle = 90.
Cheers
Paulo
Please Log in or Create an account to join the conversation.
- flaviyan
-

- Offline
- Senior Boarder
- Posts: 54
The first warning can be ignored the simulations will not give you any errors or crashes. The second warning can be corrected if you follow what the comment says i.e increase tau_t to 1.0. Although it is recommended to use V-Rescale thermostat in the production runs. Hope this helps.
Please Log in or Create an account to join the conversation.
- Pim
-

- Offline
- Expert Boarder
- Posts: 105
James Starlight wrote: Thanks so much for the suggestions, Riccardo!
However in my case (not polarize ff) in 80% of cases (8 runs from 10) the smaller water soluble protein find the bigger one (membrane receptor) very quick being located initially on 10 A distance await from it and establish with it strong contact kept complex until the MD!
Could we assume in this case that just rot-trans diffusion of water-soluble protein is the only what is responsible for the recognition? and the robust of complex is mainly due to the short-ranged effect (vdw strong attraction and local salt-bridges established upon binding).
Is it reasonable to track that hypothesis further to perform simulations with polarize water and compare the results?
Thanks for help!
J.
As shown in your previous posts the cutoff for your interactions is at 1.2 nm. Typically, when we do an association simulation we put the two molecules outside each others range, so I would go for at least 1.5 nm (you use 10 A or 1.0 nm, which is thus a bit small), in a comparable study we went up to 3.0 nm. With 3 nm distance you can assume some rotational diffusion, but with 1.0 nm I would say the result is very dependent on the way you originally orient the two proteins. Additionally, your approach is very similar to a docking simulation such as the DAFT approach ( pubs.acs.org/doi/full/10.1021/ct5010092 ). Here 500 (not 10!) simulatons from random orientations 3 nm apart are performed to get reasonable statistics on the binding mode of the protein.
The fact that you see that the complexes are stable is not a surprise, often protein association can be slightly overestimated compared to dissociation. The polarisable force field is probably not going to change very much about that: the polarisable force field will make salt-bridges potentially stronger as the charges can come closer together.
I suggest you to read the DAFT paper (again) and use a similar approach to determine the principal components or density maps of binding.
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
Sorry, I put the mistake the actual distance is much bigger - initially I start with the docked pose where both proteins are bound then I just translated smaller protein along Z on 15 A away from the bigger protein. Surprisingly even on those big distance both proteins have been associated very rapidly just for several tens of nanoseconds.
Gleb
Please Log in or Create an account to join the conversation.
- Pim
-

- Offline
- Expert Boarder
- Posts: 105
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
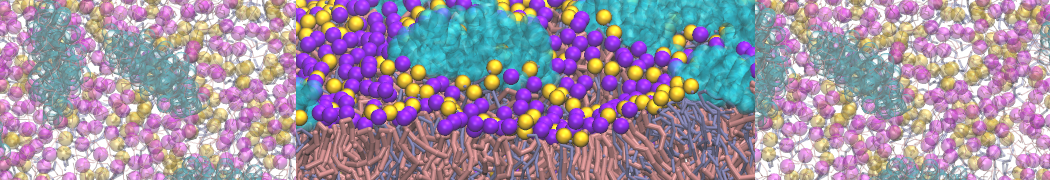
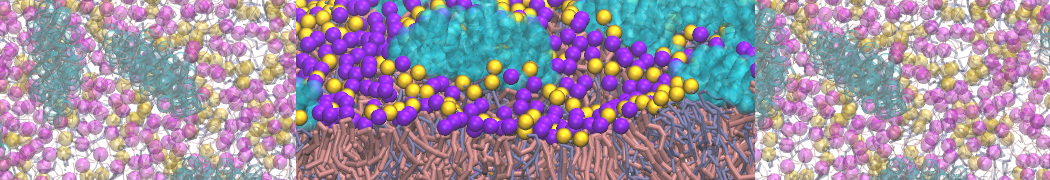
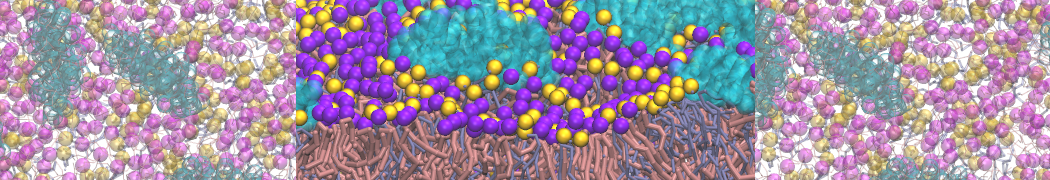
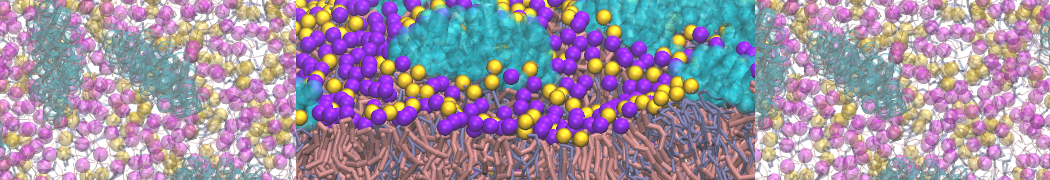
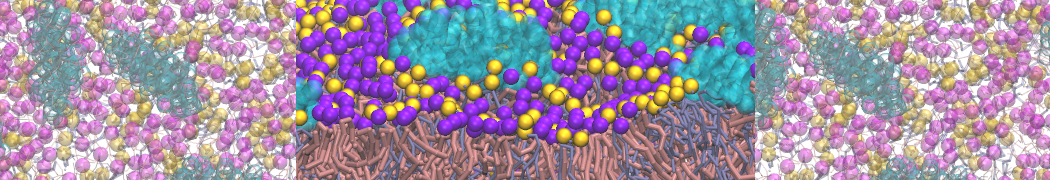
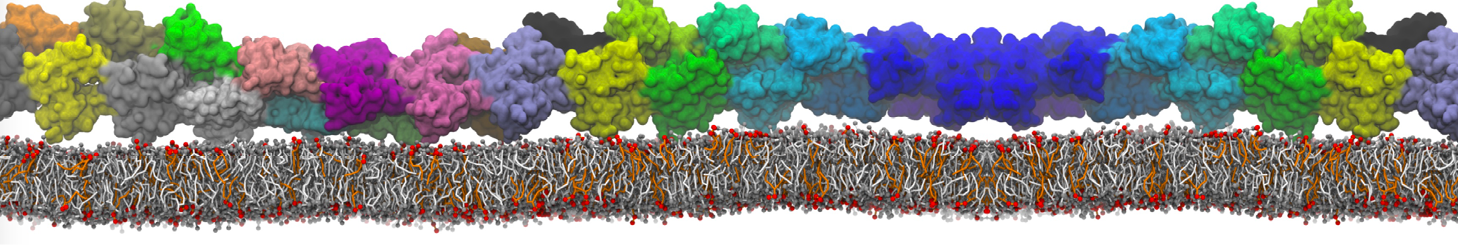
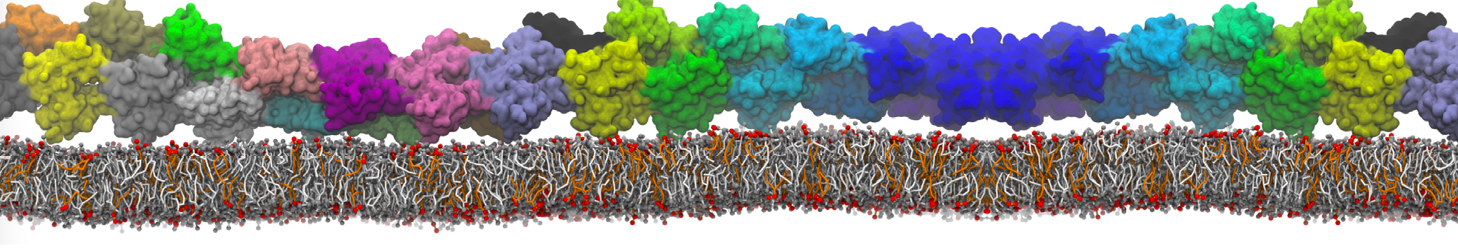
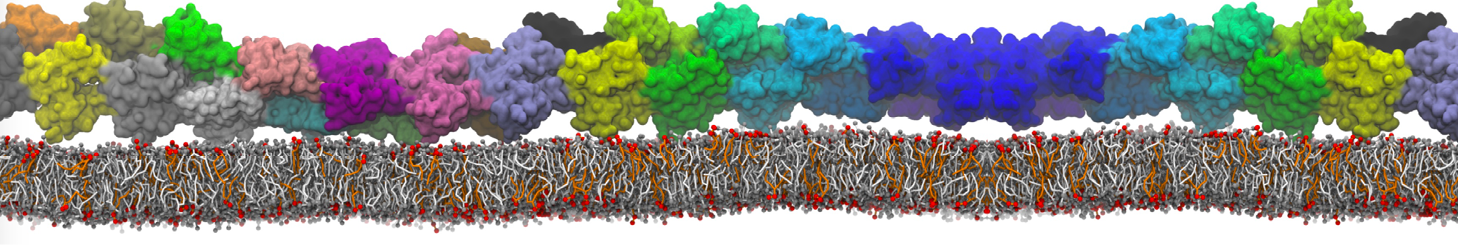
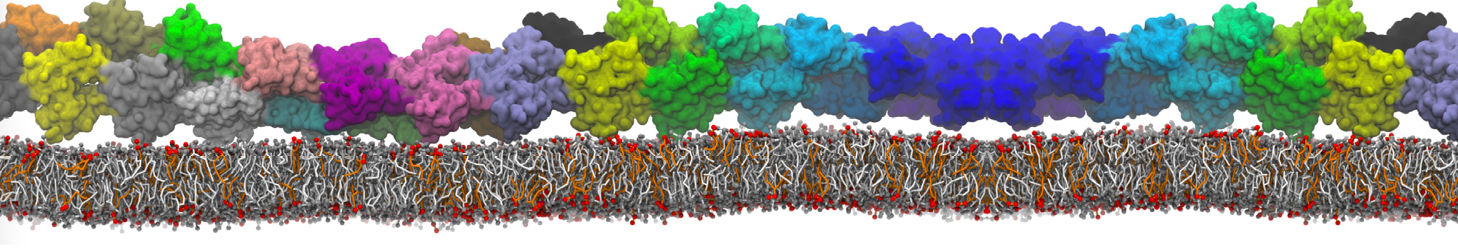
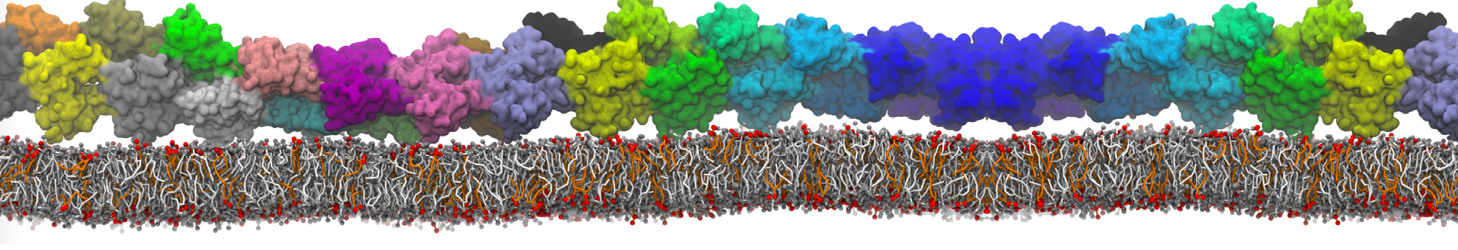
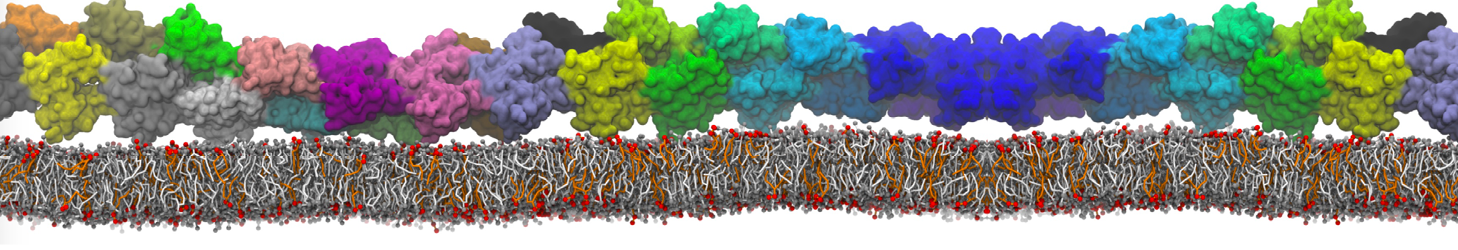
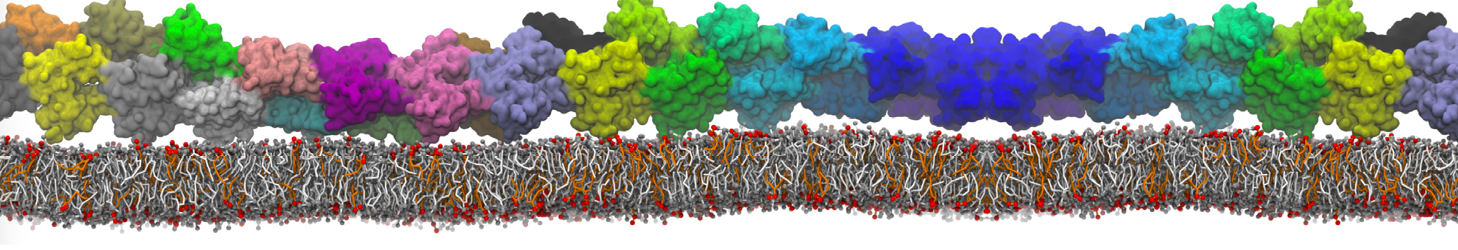
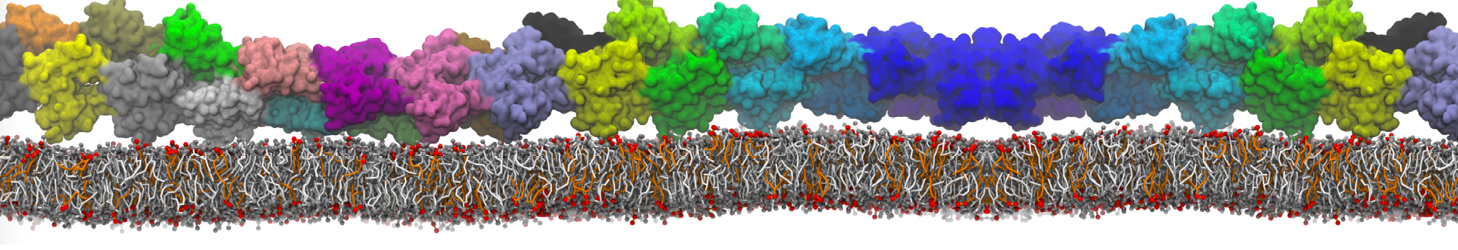
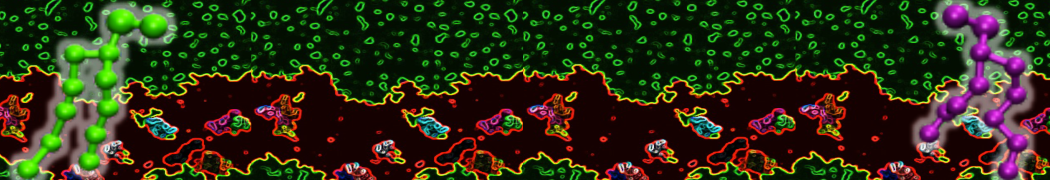
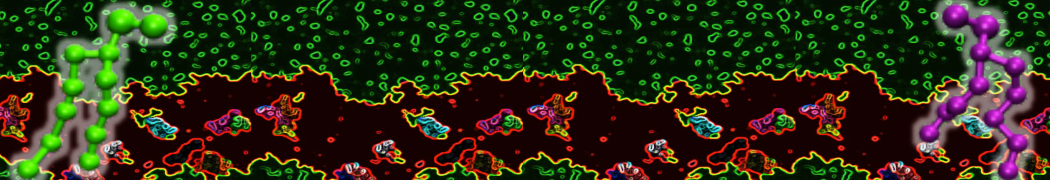
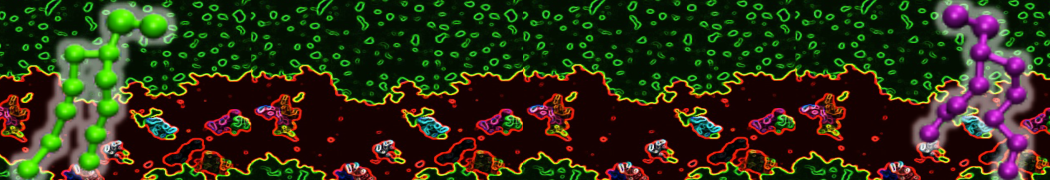
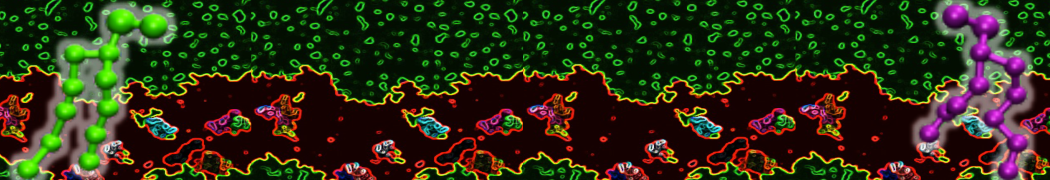
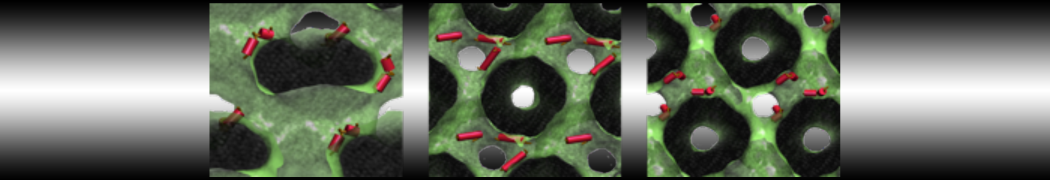
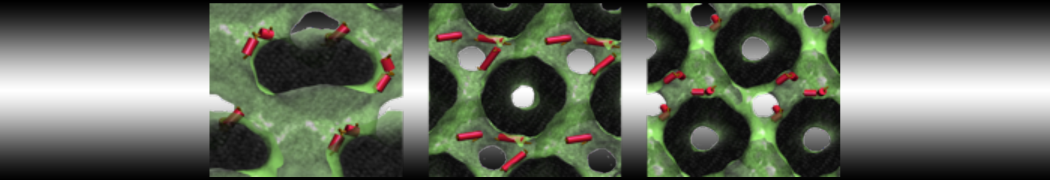
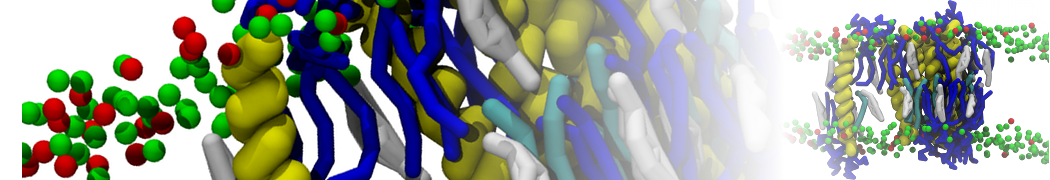
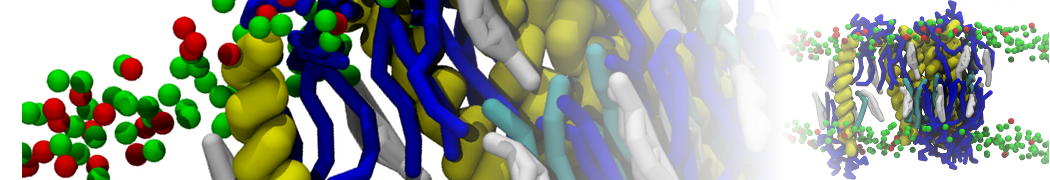
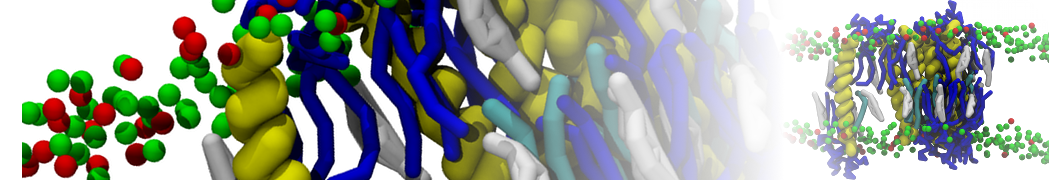
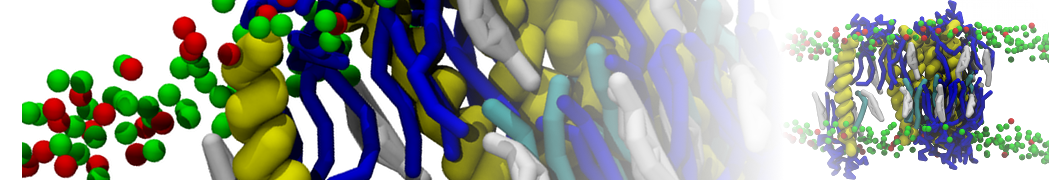
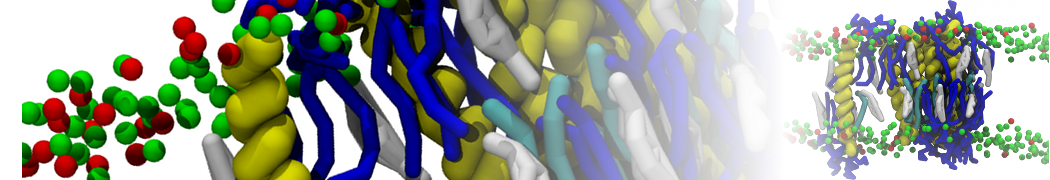
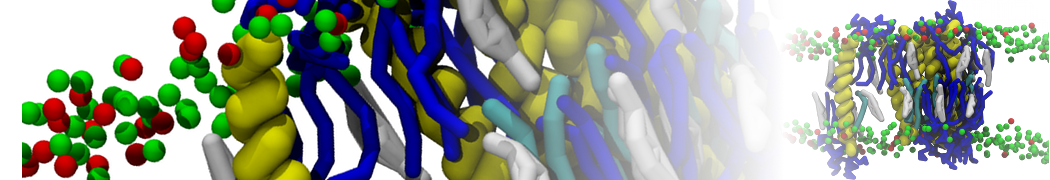
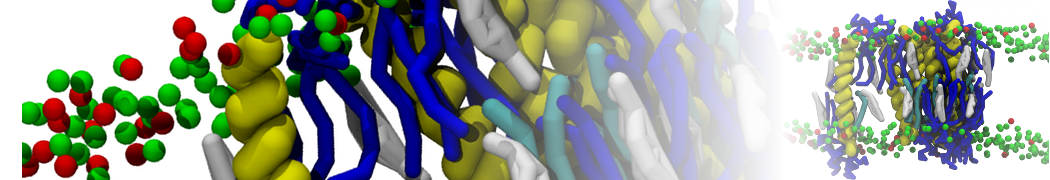
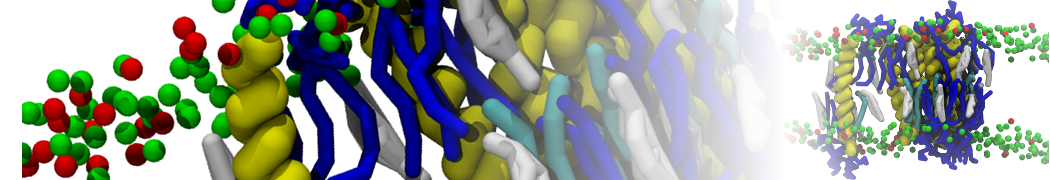
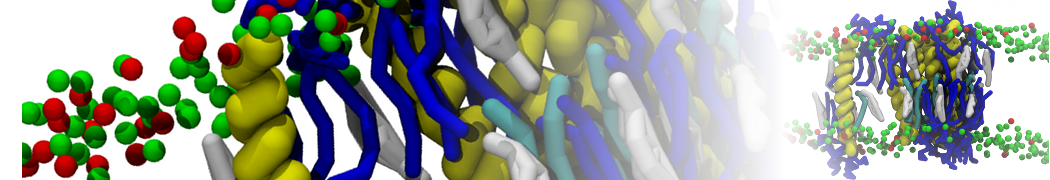
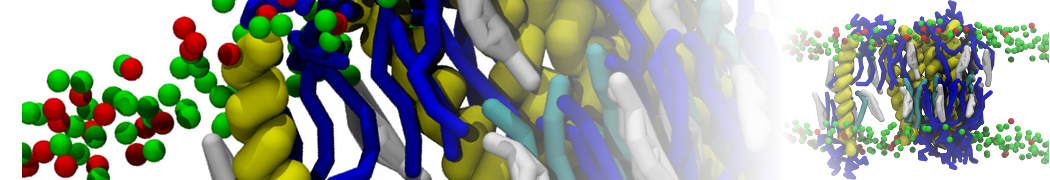
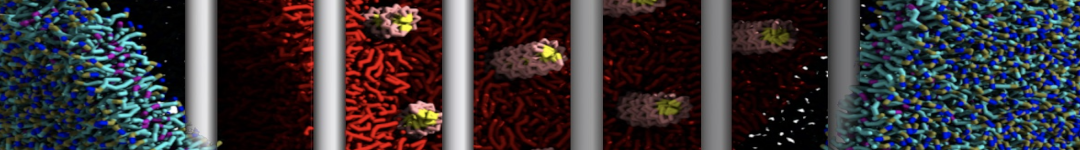
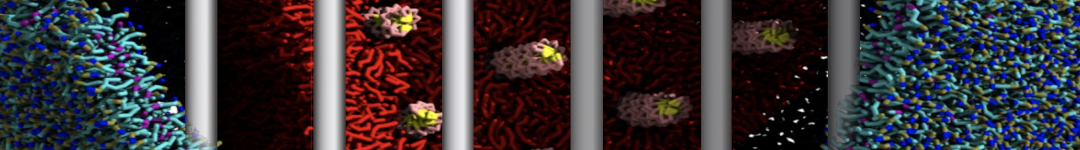
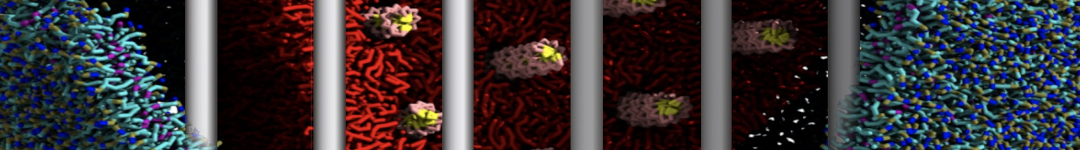
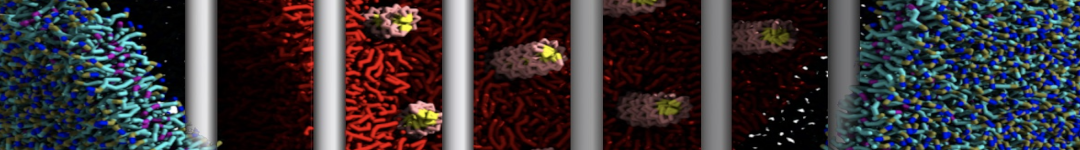
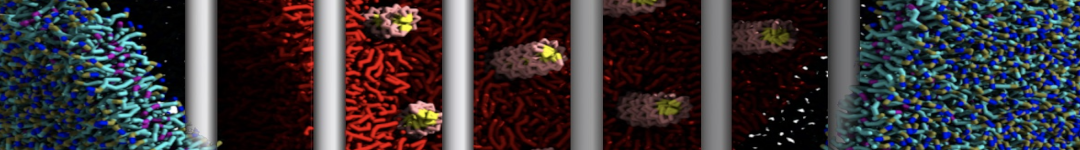
Here I started always from the same positions of both proteins
This image is hidden for guests.
Please log in or register to see it.
changing only initial velocities in each of the run assigned during its equilibration phase.
however just checked precisely the actual statistics of the association is 3 (binding established) vs 7 (not established)
also would like to repeat it with the polarizable elnydyn22 ff to see whether it mprove or not statistically the binding which might provide clues regarding significance of the electrostatics in this system :-)
Please Log in or Create an account to join the conversation.
- Pim
-

- Offline
- Expert Boarder
- Posts: 105
Please Log in or Create an account to join the conversation.
- James Starlight
-
 Topic Author
Topic Author
- Visitor
Please Log in or Create an account to join the conversation.