- Posts: 272
 Protein in water using Elnedyn
Protein in water using Elnedyn
- bt12d026
-
 Topic Author
Topic Author
- Visitor
I am doing a CG simulation of Protein in Water with Elnedyn22 ff. I am getting the following warning when I try to run my simulation under NVT using position restraints on all the beads of protein.
WARNING 1 : The bond in molecule-type Protein between atoms 1034 BB and 1035 SC1 has an estimated oscillational period of 1.1e-01 ps, which is less than 5 times the time step of 2.5e-02 ps. The notified beads in the warning belongs to HIS residue. I went through the mailing list and found some results with suggestions to ignore the warning. I have done the same but my simulation is terminating with a LINCS error. I am using GROMACS-4.5.5. My mdp file options are title = Martini define = -DPOSRES -DPOSRES_LIG integrator = md dt = 0.025 nsteps = 2000000 nstcomm = 10 comm-grps = System nstxout = 0 nstvout = 0 nstfout = 0 nstlog = 1000 nstenergy = 100 nstxtcout = 1000 xtc_precision = 100 xtc-grps = System energygrps = Protein CE PE nstlist = 10 ns_type = grid pbc = xyz rlist = 1.4 coulombtype = Shift ;Reaction_field (for use with Verlet-pairlist) ;PME (especially with polarizable water) rcoulomb_switch = 0.0 rcoulomb = 1.2 epsilon_r = 15 ; 2.5 (with polarizable water) vdw_type = Shift ;cutoff (for use with Verlet-pairlist) rvdw_switch = 0.9 rvdw = 1.2 ;1.1 (for use with Verlet-pairlist) tcoupl = v-rescale tc-grps = Protein_CE_PE W_ION tau_t = 1.0 1.0 ref_t = 310 310 pcoupl = no gen_vel = yes gen_temp = 310 gen_seed = 473529 constraints ;Pcoupltype = isotropic ;tau_p = 1.0 ;compressibility = 3e-4 ;ref_p = 1.0 = none constraint_algorithm = Lincs unconstrained_start = no lincs_order = 4 lincs_warnangle = 30 Thanks a lot[file elnedyn.top, line 30]:
The bond in molecule-type Protein between atoms 1034 BB and 1035 SC1 has an estimated oscillational period of 1.1e-01 ps, which is less than 5 times the time step of 2.5e-02 ps.
The notified beads in the warning belongs to HIS residue. I went through the mailing list and found some results with suggestions to ignore the warning. I have done the same but my simulation is terminating with a LINCS error. I am using GROMACS-4.5.5. My mdp file options are
title = Martini
define = -DPOSRES -DPOSRES_LIG
integrator = md
dt = 0.025
nsteps = 2000000
nstcomm = 10
comm-grps = System
nstxout = 0
nstvout = 0
nstfout = 0
nstlog = 1000
nstenergy = 100
nstxtcout = 1000
xtc_precision = 100
xtc-grps = System
energygrps = Protein CE PE
nstlist = 10
ns_type = grid
pbc = xyz
rlist = 1.4
coulombtype = Shift ;Reaction_field (for use with Verlet-pairlist) ;PME (especially with polarizable water)
rcoulomb_switch = 0.0
rcoulomb = 1.2
epsilon_r = 15 ; 2.5 (with polarizable water)
vdw_type = Shift ;cutoff (for use with Verlet-pairlist)
rvdw_switch = 0.9
rvdw = 1.2 ;1.1 (for use with Verlet-pairlist)
tcoupl = v-rescale
tc-grps = Protein_CE_PE W_ION
tau_t = 1.0 1.0
ref_t = 310 310
pcoupl = no
gen_vel = yes
gen_temp = 310
gen_seed = 473529
constraints ;Pcoupltype = isotropic
;tau_p = 1.0
;compressibility = 3e-4
;ref_p = 1.0 = none
constraint_algorithm = Lincs
unconstrained_start = no
lincs_order = 4
lincs_warnangle = 30
Thanks a lot
Please Log in or Create an account to join the conversation.
- djurre
-

- Offline
- Admin
Please Log in or Create an account to join the conversation.
- bt12d026
-
 Topic Author
Topic Author
- Visitor
So, what I should do? Shall I reduce the mass of that particular side chain? If so, how much?
Please Log in or Create an account to join the conversation.
- djurre
-

- Offline
- Admin
- Posts: 272
Please Log in or Create an account to join the conversation.
- bt12d026
-
 Topic Author
Topic Author
- Visitor
I have checked the martini_v2.2.itp file and found only the following section has masses below 72. So, can I change all of them to 72. If so, will it create any discrepancies in my system??
; RING types, 2-3:1 mapping
SP5 45.0 0.000 A 0.0 0.0
SP4 45.0 0.000 A 0.0 0.0
SP3 45.0 0.000 A 0.0 0.0
SP2 45.0 0.000 A 0.0 0.0
SP1 45.0 0.000 A 0.0 0.0
SNda 45.0 0.000 A 0.0 0.0
SNd 45.0 0.000 A 0.0 0.0
SNa 45.0 0.000 A 0.0 0.0
SN0 45.0 0.000 A 0.0 0.0
SC5 45.0 0.000 A 0.0 0.0
SC4 45.0 0.000 A 0.0 0.0
SC3 45.0 0.000 A 0.0 0.0
SC2 45.0 0.000 A 0.0 0.0
SC1 45.0 0.000 A 0.0 0.0
SQda 45.0 0.000 A 0.0 0.0
SQd 45.0 0.000 A 0.0 0.0
SQa 45.0 0.000 A 0.0 0.0
SQ0 45.0 0.000 A 0.0 0.0
Please Log in or Create an account to join the conversation.
- djurre
-

- Offline
- Admin
- Posts: 272
Please Log in or Create an account to join the conversation.
- bt12d026
-
 Topic Author
Topic Author
- Visitor
I have changed the masses accordingly. But no profit!!! Its been giving the same error.
Please Log in or Create an account to join the conversation.
- djurre
-

- Offline
- Admin
- Posts: 272
Please Log in or Create an account to join the conversation.
- bt12d026
-
 Topic Author
Topic Author
- Visitor
Please Log in or Create an account to join the conversation.
- xavier
-

- Offline
- Admin
- Posts: 416
You have a warning telling that a histidine BB-SC bond has a too fast vibration to be able to be described by the time step.
Your time step is 25 fs! ElNeDyn has never been able to run with more than 20 fs time step. At least with me ... try to reduce the time step to 20 fs and increase the masses and let see what happens.
bt12d026 wrote: Thank you sir for your valuable suggestions. I will try and comeback to you.
Please Log in or Create an account to join the conversation.
- bt12d026
-
 Topic Author
Topic Author
- Visitor
Sorry for the delay in reply. I have changed the time step to 20fs but no positive result yet. Still terminating with a LINCS warning. Is there any other option other than converting the constraints between backbone beads as suggested by djurre ?
Thanks a lot
Please Log in or Create an account to join the conversation.
- xavier
-

- Offline
- Admin
- Posts: 416
What type of residue is making trouble?
bt12d026 wrote: Dear Sir,
Sorry for the delay in reply. I have changed the time step to 20fs but no positive result yet. Still terminating with a LINCS warning. Is there any other option other than converting the constraints between backbone beads as suggested by djurre ?
Thanks a lot
Please Log in or Create an account to join the conversation.
- bt12d026
-
 Topic Author
Topic Author
- Visitor
Thanks for the prompt reply. I have changed the masses for the ring type structures from 45 to 72 as suggested by djurre. The LINCS warning has thrown out some pdb files which have exceptions. The problematic atoms include Water molecules and some residues like LEU,PRO,HIS..etc. It doesn't include a particular type of residues.
Please Log in or Create an account to join the conversation.
- xavier
-

- Offline
- Admin
- Posts: 416
Could you give the mdp file?
bt12d026 wrote: Dear Sir,
Thanks for the prompt reply. I have changed the masses for the ring type structures from 45 to 72 as suggested by djurre. The LINCS warning has thrown out some pdb files which have exceptions. The problematic atoms include Water molecules and some residues like LEU,PRO,HIS..etc. It doesn't include a particular type of residues.
Please Log in or Create an account to join the conversation.
- bt12d026
-
 Topic Author
Topic Author
- Visitor
This is my mdp file:
;
; STANDARD MD INPUT OPTIONS FOR MARTINI 2.x
; Updated 02 feb 2013 by DdJ
;
; for use with GROMACS 4.5/4.6
;
title = Martini
; TIMESTEP IN MARTINI
; Most simulations are numerically stable
; with dt=40 fs, some (especially rings and polarizable water) require 20-30 fs.
; Note that time steps of 40 fs and larger may create local heating or
; cooling in your system. Although the use of a heat bath will globally
; remove this effect, it is advised to check consistency of
; your results for somewhat smaller time steps in the range 20-30 fs.
; Time steps exceeding 40 fs should not be used; time steps smaller
; than 20 fs are also not required unless specifically stated in the itp file.
;define = -DPOSRES -DPOSRES_LIG
integrator = md
dt = 0.02
nsteps = 100000000
nstcomm = 10
comm-grps = System
nstxout = 0
nstvout = 0
nstfout = 0
nstlog = 1000
nstenergy = 100
nstxtcout = 1000
xtc_precision = 100
xtc-grps = System
energygrps = System
; NEIGHBOURLIST and MARTINI
; Due to the use of shifted potentials, the noise generated
; from particles leaving/entering the neighbour list is not so large,
; even when large time steps are being used. In practice, once every
; ten steps works fine with a neighborlist cutoff that is equal to the
; non-bonded cutoff (1.2 nm). However, to improve energy conservation
; or to avoid local heating/cooling, you may increase the update frequency
; and/or enlarge the neighbourlist cut-off (to 1.4 nm). The latter option
; is computationally less expensive and leads to improved energy conservation
nstlist = 10
ns_type = grid
pbc = xyz
rlist = 1.4
; MARTINI and NONBONDED
; Standard cut-off schemes are used for the non-bonded interactions
; in the Martini model: LJ interactions are shifted to zero in the
; range 0.9-1.2 nm, and electrostatic interactions in the range 0.0-1.2 nm.
; The treatment of the non-bonded cut-offs is considered to be part of
; the force field parameterization, so we recommend not to touch these
; values as they will alter the overall balance of the force field.
; In principle you can include long range electrostatics through the use
; of PME, which could be more realistic in certain applications
; Please realize that electrostatic interactions in the Martini model are
; not considered to be very accurate to begin with, especially as the
; screening in the system is set to be uniform across the system with
; a screening constant of 15. When using PME, please make sure your
; system properties are still reasonable.
;
; With the polarizable water model, the relative electrostatic screening
; (epsilon_r) should have a value of 2.5, representative of a low-dielectric
; apolar solvent. The polarizable water itself will perform the explicit screening
; in aqueous environment. In this case, the use of PME is more realistic.
;
; For use in combination with the Verlet-pairlist algorithm implemented
; in Gromacs 4.6 a straight cutoff in combination with the potential
; modifiers can be used. Although this will change the potential shape,
; preliminary results indicate that forcefield properties do not change a lot
; when the LJ cutoff is reduced to 1.1 nm. Be sure to test the effects for
; your particular system. The advantage is a gain of speed of 50-100%.
coulombtype = Shift ;Reaction_field (for use with Verlet-pairlist) ;PME (especially with polarizable water)
rcoulomb_switch = 0.0
rcoulomb = 1.2
epsilon_r = 15 ; 2.5 (with polarizable water)
vdw_type = Shift ;cutoff (for use with Verlet-pairlist)
rvdw_switch = 0.9
rvdw = 1.2 ;1.1 (for use with Verlet-pairlist)
;cutoff-scheme = verlet
;coulomb-modifier = Potential-shift
;vdw-modifier = Potential-shift
;epsilon_rf = 0 ; epsilon_rf = 0 really means epsilon_rf = infinity
;verlet-buffer-drift = 0.005
; MARTINI and TEMPERATURE/PRESSURE
; normal temperature and pressure coupling schemes can be used.
; It is recommended to couple individual groups in your system separately.
; Good temperature control can be achieved with the velocity rescale (V-rescale)
; thermostat using a coupling constant of the order of 1 ps. Even better
; temperature control can be achieved by reducing the temperature coupling
; constant to 0.1 ps, although with such tight coupling (approaching
; the time step) one can no longer speak of a weak-coupling scheme.
; We therefore recommend a coupling time constant of at least 0.5 ps.
; The Berendsen thermostat is less suited since it does not give
; a well described thermodynamic ensemble.
;
; Pressure can be controlled with the Parrinello-Rahman barostat,
; with a coupling constant in the range 4-8 ps and typical compressibility
; in the order of 10-4 - 10-5 bar-1. Note that, for equilibration purposes,
; the Berendsen thermostat probably gives better results, as the Parrinello-
; Rahman is prone to oscillating behaviour. For bilayer systems the pressure
; coupling should be done semiisotropic.
tcoupl = v-rescale
tc-grps = Protein_lipid W_ION
tau_t = 1.0 1.0
ref_t = 310 310
pcoupl = Parrinello-Rahman
Pcoupltype = isotropic
tau_p = 6.0 6.0
compressibility = 3e-4 3e-4
ref_p = 1.0 1.0
gen_vel = no
; MARTINI and CONSTRAINTS
; for ring systems and stiff bonds constraints are defined
; which are best handled using Lincs.
constraints = none
constraint_algorithm = Lincs
unconstrained_start = no
lincs_order = 4
lincs_warnangle = 30
Please Log in or Create an account to join the conversation.
- xavier
-

- Offline
- Admin
- Posts: 416
Then I would run a equilibration of 10 ps with small time step (1 fs) with position restrains on the protein, then repeat this with a 10 fs time and restrains only on the backbone, then switch to 20 fs time step and then release the restrains. If the problem persists we'll have to look at the topology.
How did you generate the topology? Which version of the force field did you use?
bt12d026 wrote: I have done minimization with emtol=200 kJ/mol and dt=20fs.
This is my mdp file:
;
; STANDARD MD INPUT OPTIONS FOR MARTINI 2.x
; Updated 02 feb 2013 by DdJ
;
; for use with GROMACS 4.5/4.6
;
title = Martini
; TIMESTEP IN MARTINI
; Most simulations are numerically stable
; with dt=40 fs, some (especially rings and polarizable water) require 20-30 fs.
; Note that time steps of 40 fs and larger may create local heating or
; cooling in your system. Although the use of a heat bath will globally
; remove this effect, it is advised to check consistency of
; your results for somewhat smaller time steps in the range 20-30 fs.
; Time steps exceeding 40 fs should not be used; time steps smaller
; than 20 fs are also not required unless specifically stated in the itp file.
;define = -DPOSRES -DPOSRES_LIG
integrator = md
dt = 0.02
nsteps = 100000000
nstcomm = 10
comm-grps = System
nstxout = 0
nstvout = 0
nstfout = 0
nstlog = 1000
nstenergy = 100
nstxtcout = 1000
xtc_precision = 100
xtc-grps = System
energygrps = System
; NEIGHBOURLIST and MARTINI
; Due to the use of shifted potentials, the noise generated
; from particles leaving/entering the neighbour list is not so large,
; even when large time steps are being used. In practice, once every
; ten steps works fine with a neighborlist cutoff that is equal to the
; non-bonded cutoff (1.2 nm). However, to improve energy conservation
; or to avoid local heating/cooling, you may increase the update frequency
; and/or enlarge the neighbourlist cut-off (to 1.4 nm). The latter option
; is computationally less expensive and leads to improved energy conservation
nstlist = 10
ns_type = grid
pbc = xyz
rlist = 1.4
; MARTINI and NONBONDED
; Standard cut-off schemes are used for the non-bonded interactions
; in the Martini model: LJ interactions are shifted to zero in the
; range 0.9-1.2 nm, and electrostatic interactions in the range 0.0-1.2 nm.
; The treatment of the non-bonded cut-offs is considered to be part of
; the force field parameterization, so we recommend not to touch these
; values as they will alter the overall balance of the force field.
; In principle you can include long range electrostatics through the use
; of PME, which could be more realistic in certain applications
; Please realize that electrostatic interactions in the Martini model are
; not considered to be very accurate to begin with, especially as the
; screening in the system is set to be uniform across the system with
; a screening constant of 15. When using PME, please make sure your
; system properties are still reasonable.
;
; With the polarizable water model, the relative electrostatic screening
; (epsilon_r) should have a value of 2.5, representative of a low-dielectric
; apolar solvent. The polarizable water itself will perform the explicit screening
; in aqueous environment. In this case, the use of PME is more realistic.
;
; For use in combination with the Verlet-pairlist algorithm implemented
; in Gromacs 4.6 a straight cutoff in combination with the potential
; modifiers can be used. Although this will change the potential shape,
; preliminary results indicate that forcefield properties do not change a lot
; when the LJ cutoff is reduced to 1.1 nm. Be sure to test the effects for
; your particular system. The advantage is a gain of speed of 50-100%.
coulombtype = Shift ;Reaction_field (for use with Verlet-pairlist) ;PME (especially with polarizable water)
rcoulomb_switch = 0.0
rcoulomb = 1.2
epsilon_r = 15 ; 2.5 (with polarizable water)
vdw_type = Shift ;cutoff (for use with Verlet-pairlist)
rvdw_switch = 0.9
rvdw = 1.2 ;1.1 (for use with Verlet-pairlist)
;cutoff-scheme = verlet
;coulomb-modifier = Potential-shift
;vdw-modifier = Potential-shift
;epsilon_rf = 0 ; epsilon_rf = 0 really means epsilon_rf = infinity
;verlet-buffer-drift = 0.005
; MARTINI and TEMPERATURE/PRESSURE
; normal temperature and pressure coupling schemes can be used.
; It is recommended to couple individual groups in your system separately.
; Good temperature control can be achieved with the velocity rescale (V-rescale)
; thermostat using a coupling constant of the order of 1 ps. Even better
; temperature control can be achieved by reducing the temperature coupling
; constant to 0.1 ps, although with such tight coupling (approaching
; the time step) one can no longer speak of a weak-coupling scheme.
; We therefore recommend a coupling time constant of at least 0.5 ps.
; The Berendsen thermostat is less suited since it does not give
; a well described thermodynamic ensemble.
;
; Pressure can be controlled with the Parrinello-Rahman barostat,
; with a coupling constant in the range 4-8 ps and typical compressibility
; in the order of 10-4 - 10-5 bar-1. Note that, for equilibration purposes,
; the Berendsen thermostat probably gives better results, as the Parrinello-
; Rahman is prone to oscillating behaviour. For bilayer systems the pressure
; coupling should be done semiisotropic.
tcoupl = v-rescale
tc-grps = Protein_lipid W_ION
tau_t = 1.0 1.0
ref_t = 310 310
pcoupl = Parrinello-Rahman
Pcoupltype = isotropic
tau_p = 6.0 6.0
compressibility = 3e-4 3e-4
ref_p = 1.0 1.0
gen_vel = no
; MARTINI and CONSTRAINTS
; for ring systems and stiff bonds constraints are defined
; which are best handled using Lincs.
constraints = none
constraint_algorithm = Lincs
unconstrained_start = no
lincs_order = 4
lincs_warnangle = 30
Please Log in or Create an account to join the conversation.
- bt12d026
-
 Topic Author
Topic Author
- Visitor
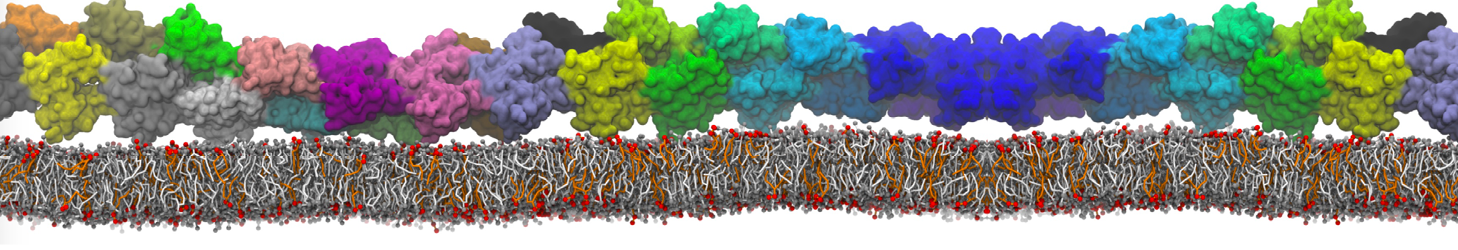
My system doesn't have a lipid membrane. My protein has some bound lipid molecules.
I have generated the topology using the command
./martinize.py -f protein.pdb -o elnedyn.top -x elnedyn.pdb -dssp /usr/local/bin/dssp -p backbone -ff elnedyn22 -ef 500 -eu 0.9
Please Log in or Create an account to join the conversation.
- xavier
-

- Offline
- Admin
- Posts: 416
bt12d026 wrote: Dear Sir,
My system doesn't have a lipid membrane. My protein has some bound lipid molecules.
I have generated the topology using the command
./martinize.py -f protein.pdb -o elnedyn.top -x elnedyn.pdb -dssp /usr/local/bin/dssp -p backbone -ff elnedyn22 -ef 500 -eu 0.9
Please Log in or Create an account to join the conversation.
- bt12d026
-
 Topic Author
Topic Author
- Visitor
Please Log in or Create an account to join the conversation.
- xavier
-

- Offline
- Admin
- Posts: 416
I suggest that you follow Djurre advice and switch the backbone constrains into bonds with large (100000) force constant as in the original ElNeDyn. You could also try to generate a topology for that original ElNeDyn with martinize.
bt12d026 wrote: I have done equilibration with 1 fs for 50ns with position restraints on entire Protein molecule then slowly released the restraints with a time step of 20fs with a simulation time of 50ns.
Please Log in or Create an account to join the conversation.